Abbott received FDA Emergency Use Authorization and launched test to detect novel Coronavirus
On Mar. 18, 2020, Abbott announced that the the U.S. Food and Drug Administration had issued Emergency Use…

On Mar. 18, 2020, Abbott announced that the the U.S. Food and Drug Administration had issued Emergency Use…

On Mar. 18, 2020, Mateon Therapeutics reported significant progress in deploying its phase 3 clinical asset, OT-101, against…

On Mar. 18, 2020, Eli Lilly announced its scientists were partnering with the Indiana State Department of Health…

On Mar. 17, 2020, Quidel Corp. announced it had received Emergency Use Authorization (EUA) from the U.S. Food…
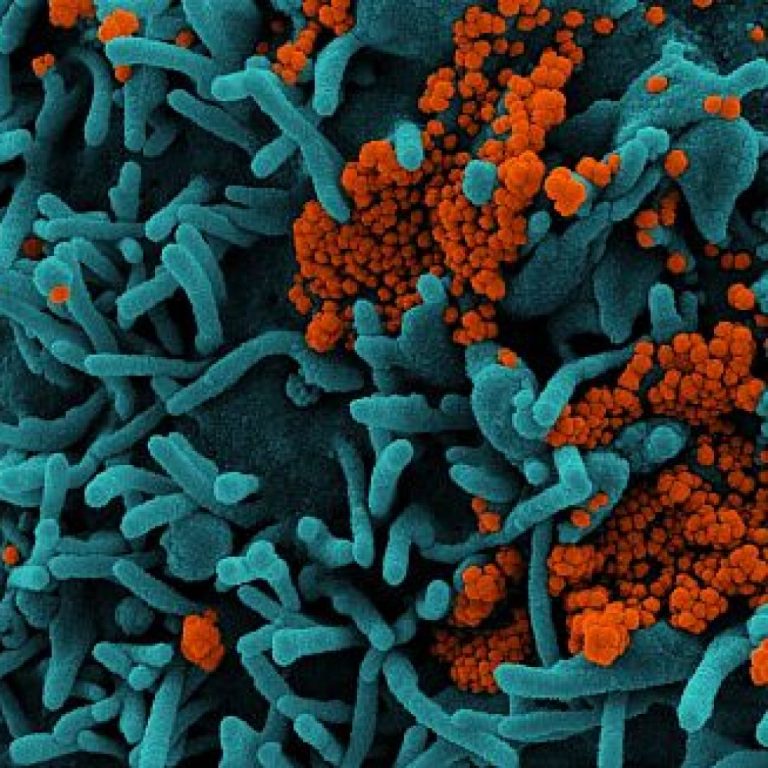
On Mar. 17, 2020, Todos Medical announced a non-exclusive distribution agreement with 3D Biomedicine Science & Technology, a…

On Mar. 16, 2020, Aimmune Therapeutics announced that the first patients in the U.S. were being treated with…

On Mar. 16, 2020, Hologic announced the U.S. Food and Drug Administration (FDA) had granted Emergency Use Authorization…

On Mar. 16, 2020, BD (Becton, Dickinson and Company) announced the companies had submitted Emergency Use Authorization requests…

On Mar. 13, 2020, Roche announced the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization…

On Mar. 13, 2020, Thermo Fisher Scientific announced the U.S. Food and Drug Administration (FDA) has issued an emergency…

On Mar. 13, 2020, declared a COVID-related emergency under Section 501(b) of the Stafford Act [18]. Under the…

On Mar. 9, 2020, The U.S. Food and Drug Administration (FDA) approved Ofev (nintedanib) oral capsules to treat…

On Mar. 6, 2020, the U.S. Food and Drug Administration (FDA) approved Isturisa (osilodrostat) oral tablets for adults…

On Mar. 6, 2020, Innovation Pharmaceuticals continues to receive inquiries regarding the potential of Brilacidin, the Company’s novel…

On Mar. 4, 2020, the UW Medicine Clinical Virology Lab announced it had received U.S. Food and Drug…
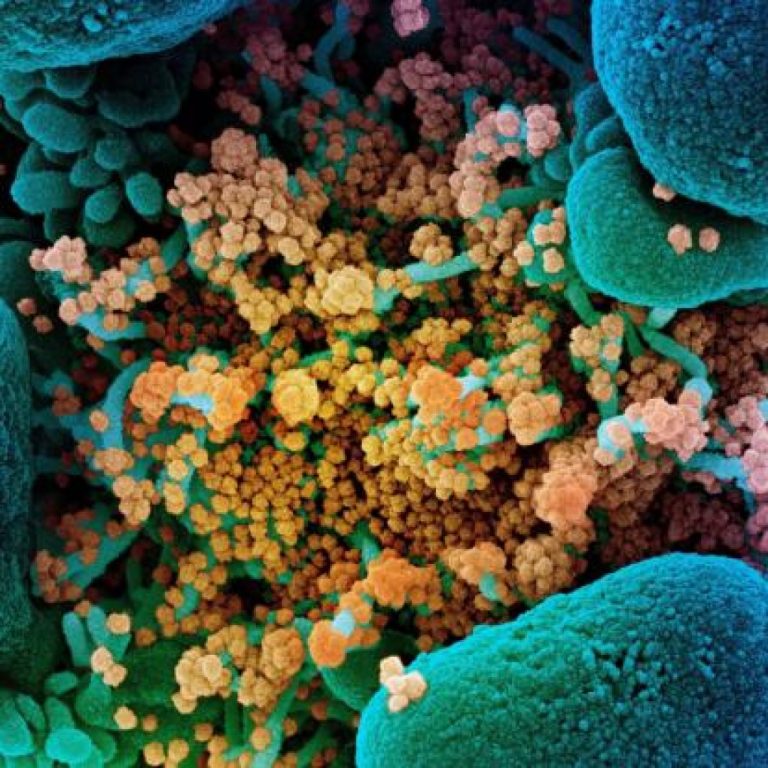
On Mar. 2, 2020, Mateon Therapeutics reported the company had been evaluating its therapeutic and AI platforms to…

On Feb. 29, 2020, the University of Washington (UW) Medicine Clinical Virology Lab announced it had received U.S….

On Feb. 28, 2020, the FDA approved an application for the first generic of Daraprim (pyrimethamine) tablets for…

On Feb. 24, 2020, Innovation Pharmaannounced the Company submitted a Material Transfer Agreement (MTA) with a leading U.S.-based…

On Feb. 22, 2020, a federal judge from the Southern District of New York ruled drug companies, device…

On Feb. 21, 2020, the U.S. Food and Drug Administration (FDA) approved NEXLETOL (bempedoic acid) tablet, an oral,…

On Feb. 21, 2020, the U.S. Food and Drug Administration (FDA) authorized marketing of the first test to…

On Feb. 18, 2020, Innovation Pharma announced the Company was exploring its lead defensin mimetic drug candidate, Brilacidin,…
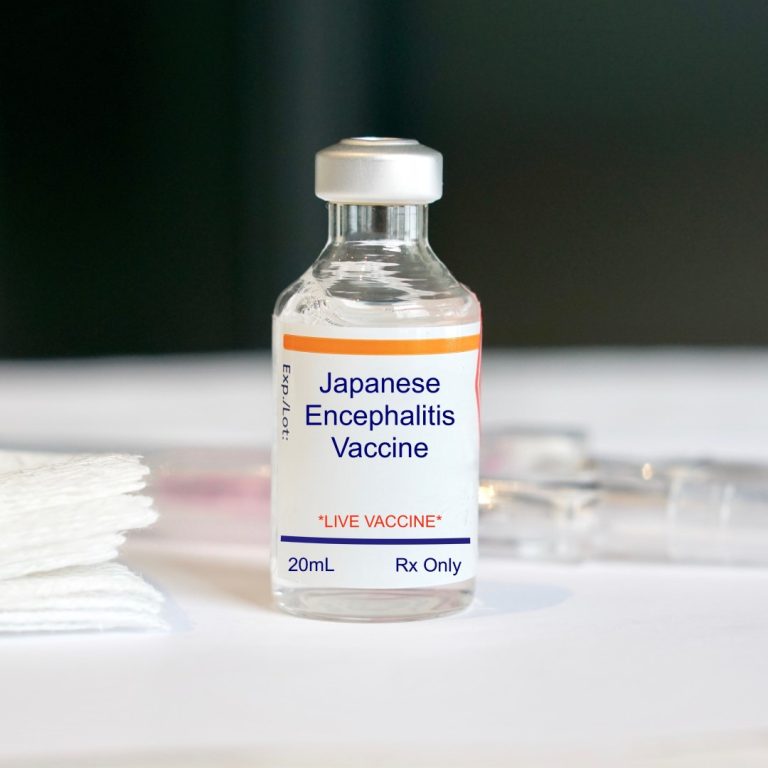
On Jan. 14, 2020, Valneva announced that the U.S. Department of Defense (DoD) had exercised an option to…

On Jan. 8, 2020, the U.S. Food and Drug Administration (FDA) approved patient-specific airway stents developed by Cleveland…

On Dec. 20, 2019, the U.S. Food and Drug Administration (FDA) granted accelerated approval to Daiichi Sankyo’s Enhertu…

On Dec. 18, 2019, Seagen and Astellas Pharma announced the U.S. Food and Drug Administration (FDA) had granted…

On Dec. 17, 2019, the University of Alabama at Birmingham Comprehensive Cancer Center (UAB) announced that Gregory Friedman,…
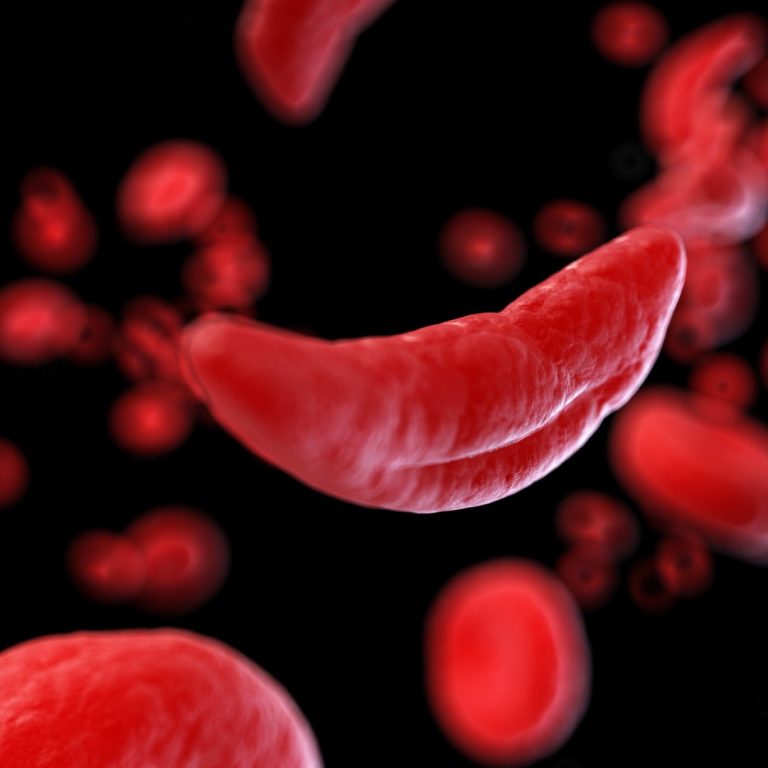
On Dec. 2, 2019, the U.S. Food and Drug Administration (FDA) announced it had approved the drug crizanlizumab…
On Nov. 22, 2019, the Centers for Disease Control and Prevention”s (CDC) Advisory Committee on Immunization Practices (ACIP)…