FDA approves first cancer drug based on Broad Institute science
On Nov. 20, 2025, the US Food and Drug Administration (FDA) has approved the first cancer drug based…

On Nov. 20, 2025, the US Food and Drug Administration (FDA) has approved the first cancer drug based…

On Nov. 20, 2025, the National Institutes of Health (NIH) has canceled funding for at least 383 clinical…

On Nov. 18, 2025, Arrowhead Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) has approved REDEMPLO…

On Nov. 17, 2025, Sandoz announced that TYRUKO® (natalizumab-sztn) is available to patients in the US. Developed by…

On Nov. 13, 2025, the U.S. Food and Drug Administration (FDA) approved multiple denosumab biosimilars, enhancing market access…

On Nov. 6, 2025, Eli Lilly announced an agreement with the U.S. government to expand access to its…

On Nov. 4, 2025, the U.S. Food and Drug Administration (FDA) approved revumenib, a first-in-class oral menin inhibitor,…

On Oct. 29, 2025, the U.S. Food and Drug Administration (FDA) announced it is aiming to reduce the…

On Oct. 28, 2025, Eli Lilly said on Tuesday it was collaborating with Nvidia to build a supercomputer…

On Oct. 27, 2025, Qureator announced a major milestone, confirming a fundamental shift in drug development under the…
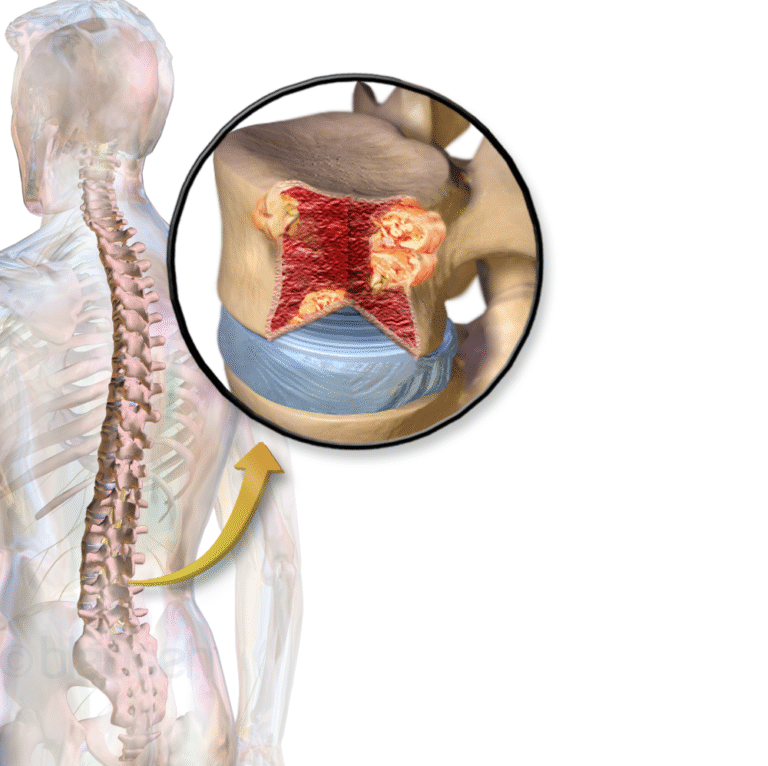
On Oct. 23, 2025, GSK announced the US Food and Drug Administration (FDA) has approved Blenrep (belantamab mafodotin-blmf)…

On Oct. 20, 2025, Glaukos announced today the U.S. Food and Drug Administration (FDA) approved its Epioxa™ HD…

On Oct. 17, 2025, Novo Nordisk announced that the US Food and Drug Administration (FDA) has approved Rybelsus®, the only oral GLP-1…

On Oct. 16, 2025, the Food and Drug Administration (FDA) announced the first round of experimental drugs that…

On Oct. 13, 2025, the U.S. Food and Drug Administration (FDA) announces it has cleared the Elecsys pTau181…

On Oct. 9, 2025, Boehringer Ingelheim’s JASCAYD® (nerandomilast) tablets has been approved by the U.S. Food and Drug…

On Oct. 9, 2025, Celltrion announced announced that the U.S. Food and Drug Administration (FDA) has approved EYDENZELT® (aflibercept-boav), biosimilar…

On Oct. 3, 2025, the U.S. Food and Drug Administration (FDA) announced a new pilot prioritization program for…
On Oct. 2, 2025, the U.S. Food and Drug Administration (FDA) approved Evita Solution’s generic version of the…

On Sept. 22, 2025, the U.S. Food and Drug Administration (FDA) announced it h initiated the approval of…

On Sept. 15, 2025, Corstasis Therapeutics announced that the U.S. Food and Drug Administration (FDA) has approved ENBUMYST™…

On Sept. 15, 2025, the U.S. Department of Agriculture (USDA) Animal and Plant Health Inspection Service (APHIS) National…

On Sept. 8, 2025, Pfizer and BioNTech announced positive topline results from an ongoing Phase 3 clinical trial…

On Sept. 4, 2025, roughly 24 hours after the launch of the West Coast Health Alliance, Hawaii is…

On Sept. 4, 2025, Novavax announced a milestone payment from Takeda has been triggered by regulatory approval for Novavax’s…
On Sept. 3, 2025, California Governor Gavin Newsom, Oregon Governor Tina Kotek, and Washington Governor Bob Ferguson announced…

On Aug. 27, 2025, Novavax announced that the U.S. Food and Drug Administration (FDA) has approved the Nuvaxovid™ 2025-2026…
On Aug. 25, 2025, shares of French drugmaker Valneva slumped more than 20% after the U.S. Food and…

On Aug. 18, 2025, Vanda Pharmaceuticals announced that it secured a landmark victory over the U.S. Food and…

On Aug. 18, 2025, Novo Nordisk announced the launch of a new offer that enables self-paying, eligible, type…