CytoDyn submited protocol with FDA for phase 3 Registrational trial of leronlimab for critically ill COVID-19 population
On Dec. 9, 2021, CytoDyn announced that it had submitted a Phase 3, randomized, double blind, placebo controlled…

On Dec. 9, 2021, CytoDyn announced that it had submitted a Phase 3, randomized, double blind, placebo controlled…

On Dec. 9, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had expanded…

On Dec. 5, 2021, Roche announced that its planned to launch the SARS-CoV-2 & Flu A/B Rapid Antigen…
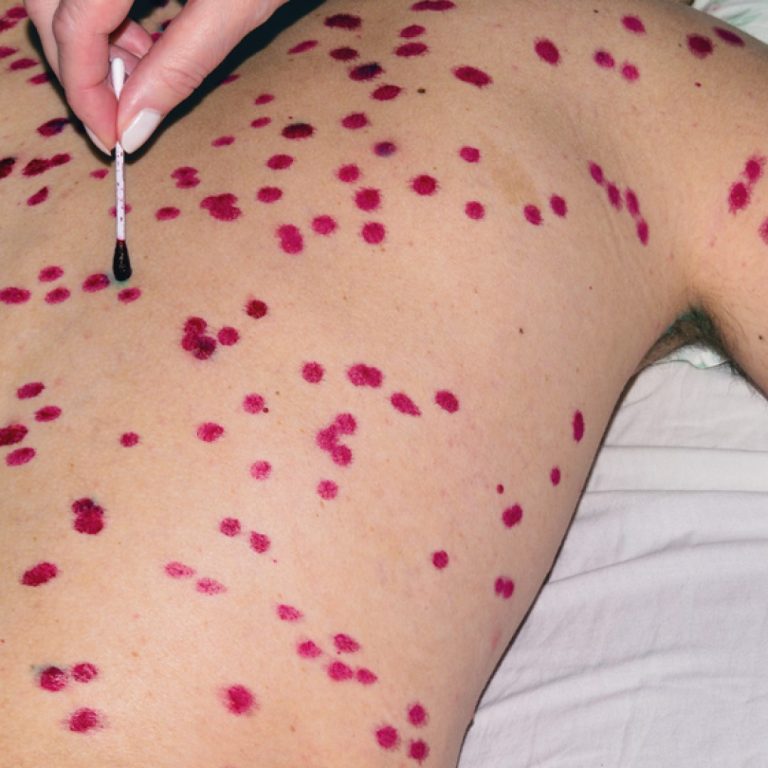
On Dec. 1, 2021, SIGA Technologies announced that Health Canada had approved oral TPOXX (tecovirimat) as an extraordinary…
On Nov. 30, 2021, Inovio Pharma announced the company was rapidly moving to evaluate its COVID-19 DNA vaccine…

On Nov. 24, 2021, Johnson & Johnson announced the U.S. Food and Drug Administration (FDA) had issued Emergency…

On Nov. 22, 2021, the U.S. Food and Drug Administration (FDA) authorized another over-the-counter (OTC) COVID-19 antigen test. The…

On Nov. 19, 2021, the U.S. Food and Drug Administration (FDA) announced that it had extended the emergency…

On Nov. 19, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had expanded…
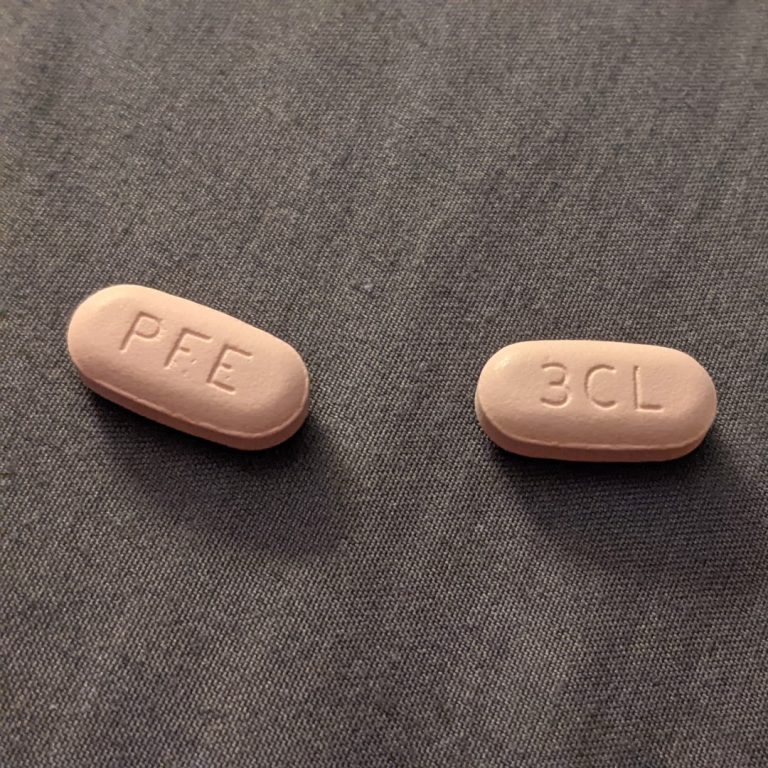
On Nov. 18, 2021, Pfizer announced an agreement with the U.S. government to supply 10 million treatment courses…

On Nov. 17, 2021, Zosano Pharma announced that the Philippine Food and Drug Administration had granted emergency use…

On Nov. 17, 2021, GlaxoSmithKline and Vir Biotechnology announced U.S. government contracts totalling approximately $1 billion (USD) to…

On Nov. 16, 2021, Pfizer announced it had submitted an Emergency Use Authorization (EUA) of its investigational oral…

On Nov. 11, 2021, Innovation Pharma reported topline results from the Companyメs Phase 2 clinical trial of Brilacidin…

On Nov. 10, 2021, Meridian Bioscience announced that their Revogeneᆴ SARS-CoV-2 assay was granted Emergency Use Authorization by…

On Nov. 9, 2021, Moderna announced that it has submitted for a variation to the conditional marketing authorization…

On Nov. 9, 2021, Inovio Pharma announced that the U.S. Food and Drug Administration (FDA) provided authorization to…

On Nov. 8, 2021, The U.S. Food and Drug Administration (FDA) reissued the emergency use authorization (EUA) for…

On Nov. 5, 2021, the U.S. Food and Drug Administration The FDA issued an emergency use authorization (EUA)…
On Nov. 2, 2021, OraSure Technologies announced that the EUA for its InteliSwabル COVID-19 rapid tests had been…

On Nov. 1, 2021, the U.S. Food and Drug Administration (FDA) cleared the first 510(k) for a COVID-19…

On Oct. 31, 2021, Moderna announced that the U.S. Food and Drug Administration (FDA) had notified the Company…
On Oct. 29, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had authorized…
On Oct. 26, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administrationメs (FDA) Vaccines and…

On Oct. 25, 2021, Moderna announced positive interim data from the Phase 2/3 study, called the KidCOVE study,…

On Oct. 20, 2021, Moderna announced that the U.S. Food and Drug Administration (FDA) had authorized for emergency…

On Oct. 20, 2021, Johnson & Johnson announced the U.S. Food and Drug Administration (FDA) had issued Emergency…
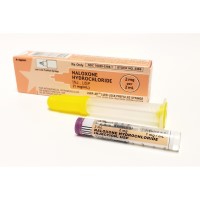
On Oct. 18, 2021, the Food and Drug Administration approved ZIMHI (naloxone hydrochloride) injection as an additional option…

On Oct. 18, 2021, Gilead Sciences announced that the Food and Drug Administration had approved a new low-dose…

On Oct. 15, 2021, Roche announced that the U.S. Food and Drug Administration (FDA) had approved Tecentriq (atezolizumab)…