Veru submitted EUA to U.S. FDA for Sabizabulin, its anti-Inflammatory drug candidate for hospitalized COVID-19 patients
On Jun. 7, 2022, Veru announced that is had submitted an emergency use authorization (EUA) application to the…
On Jun. 7, 2022, Veru announced that is had submitted an emergency use authorization (EUA) application to the…

On Apr. 28, 2022, Moderna announced that it had submitted a request for emergency use authorization (EUA) for…

On Apr. 26, 2022, Pfizer and BioNTech submitted an application to the U.S. Food and Drug Administration (FDA)…

On Apr. 25, 2022, Gilead Sciences announced that the U.S. Food and Drug Administration (FDA) has approved a supplemental…

On Nov. 12, 2021, Regeneron announced that the U.S. Food and Drug Administration had extended by three months…

On Apr. 8, 2022, Novavax announced and Serum Institute of India (SII), the world’s largest vaccine manufacturer by…

On Mar. 31, 2022, Sorrento Therapeutics announced that the FDA had given clearance to commence the Phase 3…

On Mar. 29, 2022, Moderna announced that it had received approval from the U.S. Food and Drug Administration…
On Mar. 29, 2022, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had expanded…

On Mar. 25, 2022, GlaxoSmithKline and Vir Biotechnology announced that the US Food and Drug Administration (FDA) had…
On Mar. 17, 2022, Moderna announced that it had submitted a request to the U.S. Food and Drug…

On Mar. 7, 2022, the U.S. Food and Drug Administration (FDA) announced it had made a low-risk determination…
On Mar. 2, 2022, Sorrento Therapeutics announced that its Phase I study of intranasal (IN) STI-9199 (COVISHIELDTM IN) has…

On Feb. 11, 2022, Pfizer and BioNTech announced plans to extend their rolling submission to the U.S. Food…

On Feb. 1, 2022, Pfizer and BioNTech announced that following a request from the U.S. Food and Drug…

On Jan. 31, 2022, Moderna announced the U.S. Food and Drug Administration (FDA) had approved the Biologics License…

On Jan. 31, 2022, Novavax announced that it had submitted a request to the U.S Food and Drug…
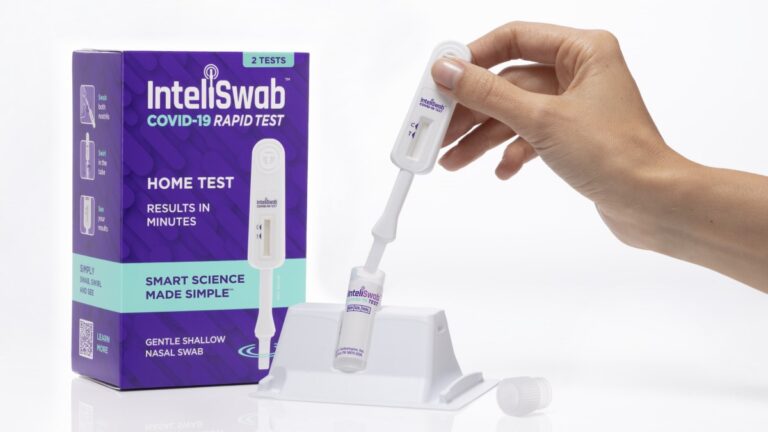
On Jan. 31, 2022, OraSure Technologies announced that its InteliSwab COVID-19 rapid tests had been authorized by the…

On Jan. 31, 2022, Veru announced that the U.S. Food and Drug Administration (FDA) had granted Fast Track…

On Jan. 26, 2022, the NIH announced that adults who had previously received a full regimen of any…
On Jan. 20, 2022, Gilead Sciences announced that the U.S. Food and Drug Administration had granted expedited approval…

On Jan. 12, 2022, Novavax and SK bioscience, a vaccine business subsidiary of Korea-based SK Group, announced that…

On Jan. 11, 2022, Pfizer and BioNTech announced that the U.S. Food and Drug Administration had expanded the…

On Jan. 10, 2022, Novartis Molecular Partners announced that Part A of the EMPATHY clinical trial that compared…

On Jan 5, 2022, NRx Pharmaceuticals announced that it had submitted an application for Emergency Use Authorization (EUA)…

On Jan. 3, 2022, Pfizer and BioNTech announced that the U.S. Food and Drug Administration had expanded the…

On Dec. 27, 2021, the U.S. Food and Drug Administration authorized an over-the-counter (OTC) COVID-19 antigen test, the…

Our Heroes and Remembrance illustration has Maurice Ralph Hilleman and John Enders, pioneering developers of common vaccines, and…

On Dec. 23, 2021, Merck and Ridgeback Biotherapeutics announced that the U.S. Food and Drug Administration (FDA) had…

On Dec. 22, 2021, Pfizer announced that the U.S. Food and Drug Administration (FDA) had authorized the emergency…