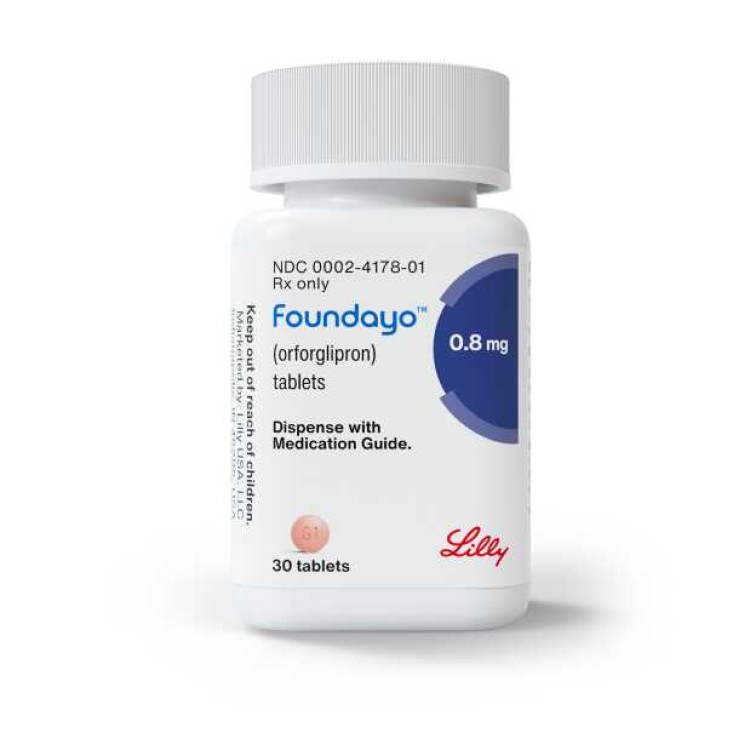
FDA approves Lilly’s Foundayo™ (orforglipron), the only GLP-1 pill for weight loss that can be taken any time of day without food or water restrictions
On Apr. 1, 2026, Eli Lilly announced the U.S. Food and Drug Administration (FDA) approved Foundayo™ (orforglipron) for…