MIT scientists debut a generative AI model that could create molecules addressing hard-to-treat diseases
On Nov. 25, 2025, a team of Massachusetts Institute of Technology scientists reported that AI model BoltzGen generates…

On Nov. 25, 2025, a team of Massachusetts Institute of Technology scientists reported that AI model BoltzGen generates…

On Nov. 24, 2025, a study published in the New England Journal of Medicine show that a test…

On Nov. 24, 2025, a new AI model called popEVE developed by Harvard Medical School researchers and colleagues…
On Nov. 20, 2025, an interdisciplinary research team at the University of Pittsburgh has developed wearable biosensors that…
On Nov. 19, 2025, the governing board of the Cancer Prevention and Research Institute of Texas (CPRIT) approved…

On Nov. 19, 2025, the Texas Animal Health Commission (TAHC) received confirmation of equine herpes myeloencephalopathy (EHM), the…

On Nov. 18, 2025, state officials in Utah report 84 measles cases in the state in a weekly…
On Nov. 17, 2025, in a major step forward for cancer care, researchers at ChristianaCare’s Gene Editing Institute…

On Nov. 17, 2025, a JAMA published study found that more than 74,000 people have had their lives…

On Nov. 14, 2025, the Washington State Department of Health reported that a Grays Harbor resident who was…

On Nov. 13, 2025, the World Health Organization (WHO) reported that Health authorities in Ethiopia are carrying out…

On Nov. 12, 2025, a study led by the Hebrew University of Jerusalem, and the Laboratory of Entomology…
On Nov. 12, 2025, Stanford Medicine scientists have found that the Epstein-Barr virus can convert B cells it’s…
On Nov. 7, 2025, research published today in Cell Reports Medicine reveals key biological differences that may explain why…

On Nov. 6, 2025, the Institute for Health Metrics and Evaluation (IHME) at the University of Washington School…

On Nov. 6, 2025, scientists from Oxford’s Radcliffe Department of Medicine have achieved the most detailed view yet…

On Nov. 5, 2025, a large study of adults with type 2 diabetes showed that those who consistently…
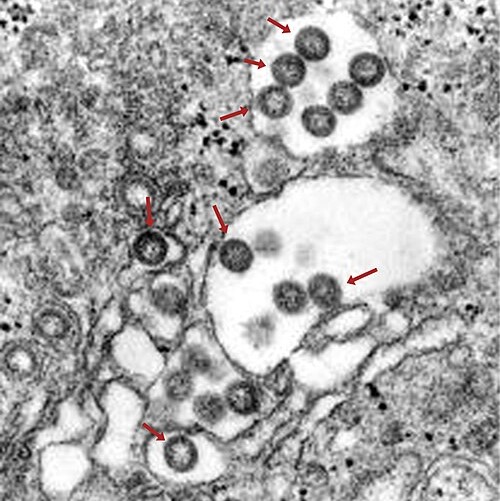
On Nov. 5, 2025, the World Health Organization (WHO) reported that between 20 September and 30 October 2025,…
On Nov. 4, 2025, six years ago this month, scientists launched a massively ambitious effort to sequence the…
On Oct. 31, 2025, investigators from Mass General Brigham, the Broad Institute of MIT and Harvard, and VA…

On Oct. 30, 2025, researchers at Virginia Commonwealth University and VCU Health announced preliminary study findings that show…
On Oct. 28, 2025, The Louisiana Illuminator reported that when there’s an outbreak of a vaccine-preventable disease, state…

On Oct. 27, 2025, researchers from Johns Hopkins Medicine and the Johns Hopkins Bloomberg School of Public Health…
As of Oct. 24, 2025, the South Carolina Department of Public Health (DPH) reported 33 cases of measles…

On. Oct. 23, 2025, Air pollution continues to exacerbate global health, including noncommunicable diseases and dementia, according to…

On Oct. 23, 2025, CareDx announced the publication of the second study from Surveillance HeartCare Outcomes Registry (SHORE)…
On Oct. 22, 2025, the South Carolina Department of Public Health (SCDPH) announced it is actively responding to…

On Oct. 21, 2025, private equity firms Blackstone and TPG said they will buy medical diagnostics firm Hologic…

On Oct. 21, 2025, the Oregon Department of Agriculture has warned that as wild birds begin to migrate…

On Oct. 18, 2025, GRAIL announced that positive performance and safety results from its registrational PATHFINDER 2 study…