Dynavax announced grant to scale up CpG 1018 adjuvant capacity to support global COVID-19 response
On Aug. 13, 2020, Dynavax Technologies announced a grant from the Bill & Melinda Gates Foundation of $3.4…

On Aug. 13, 2020, Dynavax Technologies announced a grant from the Bill & Melinda Gates Foundation of $3.4…

On Aug. 13, 2020, Novavax and SK bioscience announced a development and supply agreement for the antigen component…

On Aug. 13, 2020, Heat Biologics reported preclinical data for Heat’s gp96-based COVID-19. The data, generated at the…

On Aug. 13, 2020, the U.S. Department of Health and Human Services (HHS) announced combined investments of $6.5…

On Aug. 13, 2020, data from the WHO/UNICEF Joint Monitoring Programme (JMP) revealed that 43 per cent of…

On Aug. 12, 2020, Baxter announced it had received Emergency Use Authorizations (EUAs) from the FDA for the…

On Aug. 12, 2020, Thermo Fisher Scientific announced it had committed to supporting the efforts of historically black…

On Aug. 12, 2020, Celdara Medical announced that the NIH had awarded a Small Business Innovation Research (SBIR)…

On Aug. 11, 2020, Hologic announced that it has validated use of its Aptimaᆴ and Panther Fusionᆴ molecular…

On Aug. 11, 2020, Moderna announced that the U.S. government had secured 100 million doses of mRNA-1273. The…

On Aug. 11, 2020, LabCorpᆴ announced details of a no charge antibody testing program in response to federal…

On Aug. 10, 2020, Gilead Sciences announced that it had submitted a New Drug Application (NDA) to the…

On Aug. 10, 2020, Vaxart announced that its COVID-19 Investigational New Drug (IND) application has been filed with…

On Aug. 10, 2020, CBRᆴ by Generate Life Sciences, the worldメs largest private newborn stem cell bank, and…

On Aug. 10, 2020, Humanigen announced that the Brazilian regulatory agency, Anvisa, had granted permission to commence a…

On Aug. 10, 2020, Omeros reported the results of a compassionate-use study evaluating narsoplimab, Omerosメ investigational human monoclonal…

On Aug. 9, 2020, OpGen announced that it had commenced marketing and promotion, on a non-exclusive basis, of…
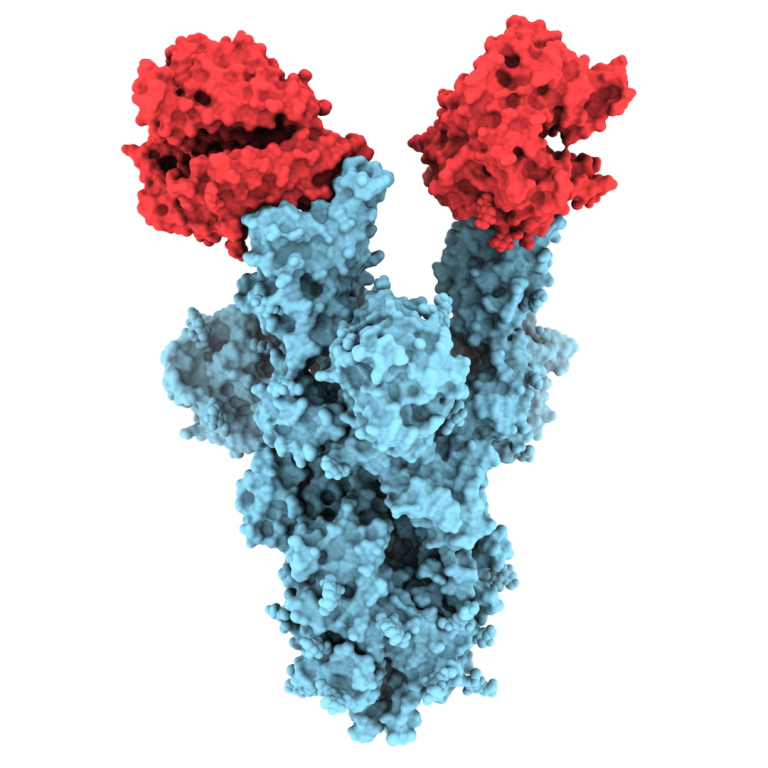
On Aug. 7, 2020, scientists at Scripps Research announced they had obtained high-resolution, atomic-scale details of the structure…
On Aug. 7, 2020, Novavax and Takeda Pharmaceutical announced a partnership for the development, manufacturing and commercialization of…

On Aug. 7, 2020, Pfizer announced a multi-year agreement with Gilead Sciences to manufacture and supply Gileadメs investigational…

On Aug. 6, 2020, RedHill Biopharma announced approval from the Mexican Federal Committee for the Protection against Sanitary…

On Aug. 6, 2020, researchers at the AIDS Institute, Department of Microbiology and State Key Laboratory of Emerging…

On Aug. 6, 2020, Novavax announced it had entered a supply and license agreement with the Serum Institute…

On Aug. 6, 2020, Organicell Regenerative Medicine announced that the FDA had approved two outpatient Emergency Investigational New…

On Aug. 5, 2020, a randomized, controlled clinical trial evaluating the safety and efficacy of a treatment regimen…

On Aug. 5, 2020, the National Institutes of Health (NIH) announced it had launched the Medical Imaging and…

On Aug. 5, 2020, the National Institute of Allergy and Infectious Diseases (NIAID) announced that the investigational vaccine…

On Aug. 5, 2020, the Tecan Group and Thermo Fisher Scientific announced a collaboration to enable scaled-up COVID-19…

On Aug. 5, 2020, BioNTech and Shanghai Fosun Pharmaceutical announced that the first 72 participants had been dosed…

On Aug. 5, 2020,Thermo Fisher Scientific announced a new highly automated, real-time PCR solution designed to analyze up…