Moderna confirmed discussions with the MHLW to supply Japan with 40 million doses of mRNA vaccine against COVID-19
On Aug. 28, 2020, Moderna confirmed that the Company was engaged in discussions with the Ministry of Health,…

On Aug. 28, 2020, Moderna confirmed that the Company was engaged in discussions with the Ministry of Health,…

On Aug. 28, 2020, iBio announced that it had entered into an exclusive worldwide license agreement with Planet…
On Aug. 27, 2020, Vaccitech announced that it had received a non-dilutive Government grant to support research with…

On Aug. 27, 2020, the U.S. Departments of Health and Human Services and the Department of and Defense…

On Aug. 27, 2020, Laurent Pharmaceuticals announced that it had received approval from the U.S. Food and Drug…

On Aug. 27, 2020, AIM ImmunoTech announced that it had identified an effective in vitro model at The…
On Aug. 27, 2020, Myriad RBM announced ultrasensitive immunoassays for viral pneumonia Including COVID-19 from our CLIA-certified laboratory….
On Aug. 27, 2020, RedHill Biopharma announced that its U.S. Phase 2 study with opaganib (Yeliva®, ABC294640) in…

On Aug. 26, 2020, UConn announced that Thanh Duc Nguyen, Assistant Professor in the Departments of Mechanical Engineering…

On Aug. 26, 2020, Altimmune announced additional positive results from the preclinical studies of its single-dose intranasal COVID-19…

On Aug. 26, 2020, Heat Biologics announced publication of positive preclinical COVID-19 results in bioRxiv. The company reported…

On Aug. 26, 2020, Abbott announced the FDA had issued Emergency Use Authorization (EUA) for its BinaxNOWル COVID-19…

On Aug. 25, 2020, AstraZeneca announced that the first participants have been dosed in a Phase I trial…

On Aug. 25, 2020, Sinovac Biotech announced it had signed two agreements in Hainan, China with PT Bio…

On Aug. 25, 2020, data from the Broad Institute of MIT and Harvard, Massachusetts General Hospital, the Massachusetts…
On Aug. 25, 2020, CytoDyn announced the Company had reached the requisite number of enrolled patients in its…
On Aug. 24, 2020, a published study led by the Universities of Oxford and Birmingham found that, compared…

On Aug. 24 2020, NantKwest and ImmunityBio announced an agreement to jointly develop, manufacture, market, and commercialize therapeutics…

On Aug. 24, 2020, Innovation Pharma reported receiving additional data from a U.S. Regional Biocontainment Laboratory (RBL) based…

On Aug. 24, 2020, humans are not the only species facing a potential threat from SARS-CoV-2, the novel…

On Aug. 24, 2020, Novavax announced that the first volunteers were enrolled in the Phase 2 portion of…

On Aug. 24, 2020, the Quest Diagnostics Foundation announced a wide-ranging initiative to address and reduce health disparities…

On Aug. 24, 2020, Catalent announced that it had contracted to provide drug substance manufacturing to AstraZeneca for…

On Aug. 24, 2020, AXIM Biotechnologies announced that it had signed an exclusive limited licensing, manufacturing and distribution…

On Aug. 24, 2020,Trevena announced that Imperial College London (ICL) had initiated a proof-of-concept study for TRV027 in…

On Aug. 24, 2020, XBiotech announced that the U.S. Food and Drug Administration (FDA) issued an emergency use…

On Aug. 23, 2020, the World Heath Organization (WHO) reported that 172 economies were engaged in discussions to…

On Aug. 23, 2020, the FDA issued an emergency use authorization (EUA) for investigational convalescent plasma for the…
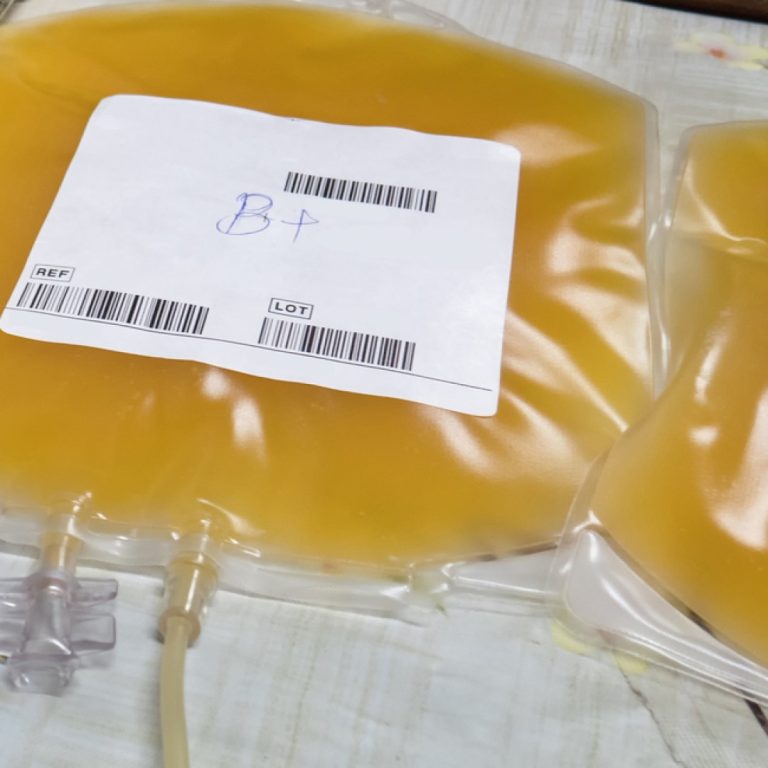
On Aug. 23, 2020, the Mayo Clinic announced that with the FDA Emergency Use Authorization of convalescent plasma,…

On Aug. 21, 2020, JAMA reported that hospitalized patients with moderate COVID-19 randomized to a 5-day course of…