bioMerieux announced CE marking of ARGENE SARS-CoV-2 diagnostic test to include saliva specimens
On Nov. 16, 2020, bioMerieux announced the expansion of its ARGENE range for the detection of SARS-CoV-2. As…
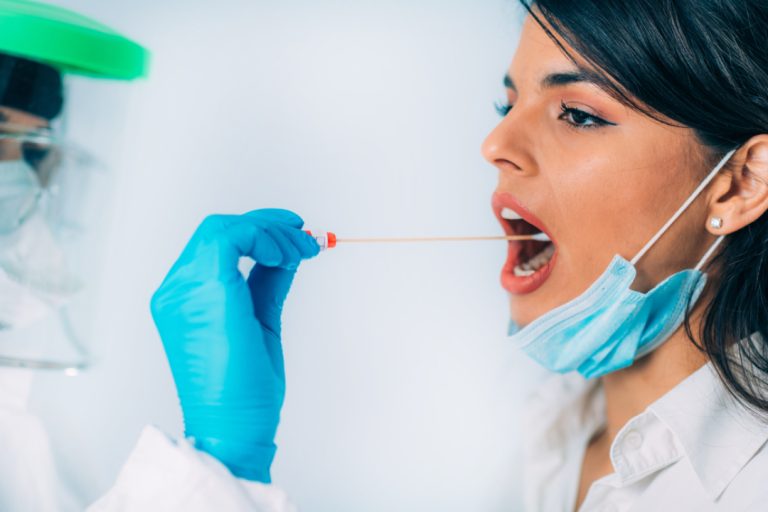
On Nov. 16, 2020, bioMerieux announced the expansion of its ARGENE range for the detection of SARS-CoV-2. As…

On Nov. 16, 2020, RedHill Biopharma announced that the U.S. Phase 2 study with opaganib (Yeliva, ABC294640)1 in…

On Nov. 16, 2020, the UK Department for Health and Social Care Testing Innovation Fund announced £12.2M funding…

On Nov. 16, 2020, Biological E. Limited (BE), a Hyderabad-based vaccines and pharmaceutical company, Dynavax Technologies, and Baylor…

On Nov. 15, 2020, Johnson & Johnson announced that it continued to enroll and vaccinate study participants for…
On Nov. 15, 2020, an independent data and safety monitoring board (DSMB) overseeing the Phase 3 trial of…
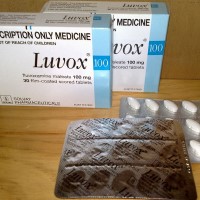
On Nov. 14, 2020, JAMA reported that in a preliminary study, adult outpatients with symptomatic COVID-19 treated with…
On Nov. 13, 2020, NeuroRx and Relief Therapeutics announced that to-date, 150 patients (out of a targeted enrollment…

On Nov. 13, 2020, Moderna announced that Swissmedic had started a rolling review of mRNA-1273, the Company’s vaccine…
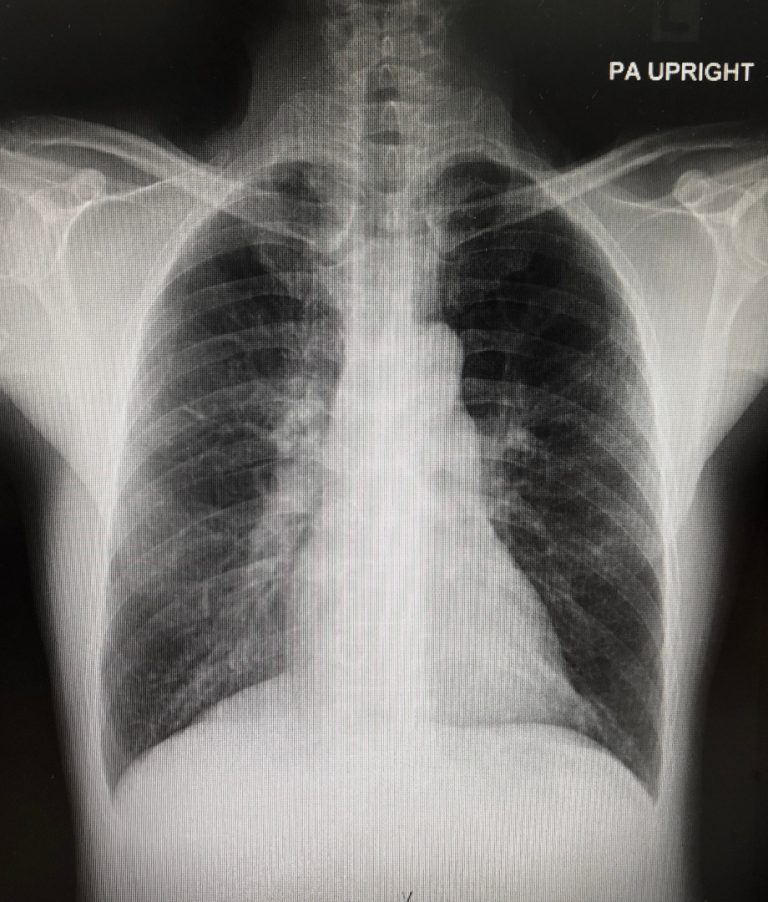
On Nov. 13, 2020, a study from Washington University School of Medicine in St. Louis and St. Jude…
On Nov. 12, 2020, Wren Laboratories announced that it had been granted an emergency use authorization by the…
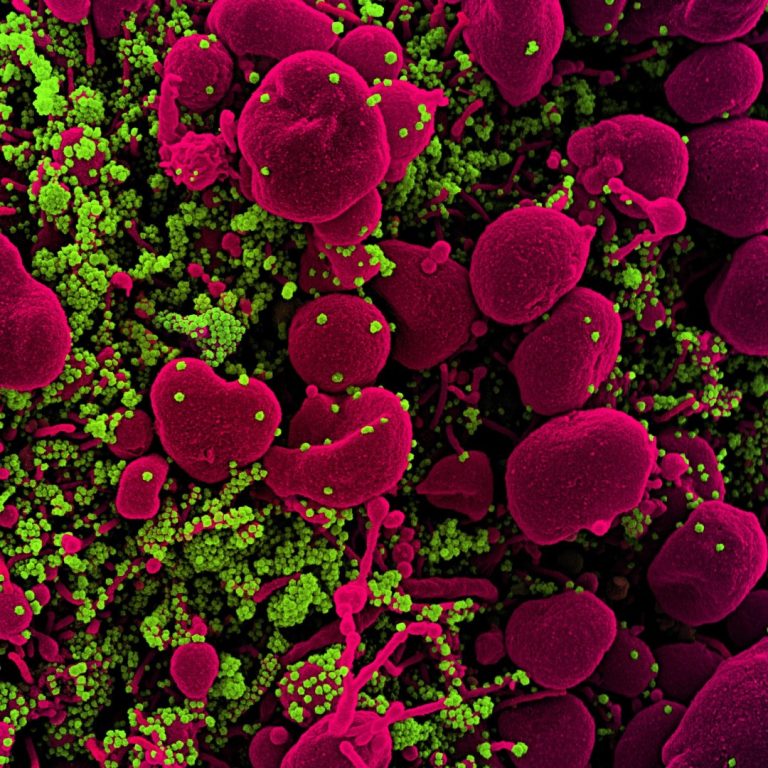
On Nov. 12, 2020, a study published in Science by a team of researchers in the U.S. and…

On Nov. 12, 2020, AstraZeneca announced that the CALAVI Phase II trials for Calquence (acalabrutinib) in patients hospitalised…

On Nov. 12, 2020, the Bill & Melinda Gates Foundation announced new commitments totaling $70 million to global…

On Nov. 11, 2020, Mesoblast announced that the randomized controlled Phase 3 trial of remestemcel-L in patients with…

On Nov. 11, 2020, Mateon Therapeutics announced an agreement with Windlas Biotech of India to commercialize ARTIVeda. ARTIVeda…

On Nov. 11, 2020, Moderna announced that it had completed case accrual for the first interim analysis of…

On Nov. 11, 2020, Cardinal Health announced it had reached an agreement with the Centers for Disease Control…

On Nov. 11, 2020, Sorrento Therapeutics announced that it was filing an investigational new drug application (IND) for…

On Nov. 11, 2020, National Institute of Allergy and Infectious Diseases (NIAID) Director Anthony S. Fauci, M.D., and…

On Nov. 10, 2020, Oxford Immunotec announced the release of data from a prospective cohort study in keyworkers…

On Nov. 19, 2020, Medigen Vaccine Biologics and BlueWillow Biologics announced a partnership to develop S-2P-NE-01, a nasal…

On Nov. 10, 2020, ImmunityBio and NantKwest, a clinical-stage, natural killer cell-based therapeutics company, announced they had administered…

On Nov. 10, 2020, Altimmune announced an agreement with Lonza for the manufacturing of AdCOVIDル, Altimmuneメs next-generation, single-dose…

On Nov. 10, 2020, 3M announced that its TB Quat Disinfectant Ready-to-Use Cleaner had been approved by the…
On Nov. 10, 2020, BD (Becton, Dickinson) announced its rapid, point-of-care, SARS-CoV-2 antigen test for use on the…
On Nov. 9, 2020, a National Institutes of Health (NIH) clinical trial evaluating the safety and effectiveness of…

On Nov. 9, 2020, a study published in JAMA reported the effect of hydroxychloroquine on clinical status at…

On Nov. 9, 2020, the FDA granted Emergency Use Authorization (EUA) for Eli Lilly’s investigational neutralizing antibody bamlanivimab…
On Nov. 9, 2020, Pfizer and BioNTech announced their mRNA-based vaccine candidate, BNT162b2, against SARS-CoV-2 had demonstrated evidence…