Medigen’s COVID-19 vaccine announced first participant dosed in phase 2 clinical trial in Taiwan
On Jan. 25, 2021, Medigen Vaccine Biologics and Dynavax Technologies announced that the first participant had been dosed…

On Jan. 25, 2021, Medigen Vaccine Biologics and Dynavax Technologies announced that the first participant had been dosed…

On Jan. 22, 2021, Aurinia Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) had approved LUPKYNISTM…

On Jan. 22, 2021, Humanigen announced an expansion to the Cooperative Research and Development Agreement (CRADA) that the…

On Jan. 22, 2021, Novavax announced that it had finalized an agreement with the Government of Canada to…

On Jan. 21, 2021, Zoetis announced it had as expanded its line of recombinant vector vaccines with the…

On Jan. 21, 2021, scientists at the Wellcome Sanger Institute announced they had created a highly detailed map…

On Jan. 21, 2021, Moderna announced that the first participant has been dosed in the Phase 1/2 study…

On Jan. 19, 2021, Cue Health announced that its molecular, point-of-care COVID-19 Tests were being distributed to five…

On Jan. 15, 2021, scientists at Washington University School of Medicine in St. Louis announced that that a…

On Jan. 14, 2021, Mammoth Biosciences announced that it had secured a subcontract with MRIGlobal, the prime contractor…

On Jan. 13, 2021, researchers announced they had developed a blood test that could make it possible for…

On Jan. 13, 2021, National Institutes of Health scientists and their colleagues announced they had identified a S….

On Jan. 13, 2021, Oxford Immunotec announced the start of a collaboration with Valneva whereby it will perform…
On Jan. 13, 2021, Mateon Therapeutics reported positive interim results from its ARTI-19 clinical trial evaluating ARTIVedaTM against…

On Jan. 13, 2021, the Oregon Department of Agriculture (ODA) continueed to test, survey, and trap at an…
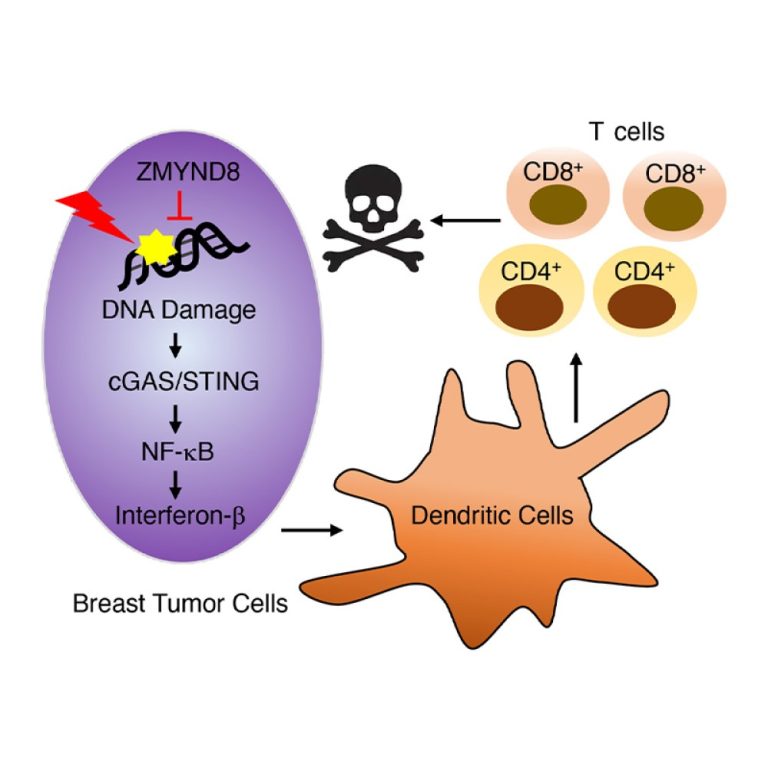
On Jan. 12, 2021, a team researchers from the University of Texas Southwestern (UTSW) announced they had identified…

On Jan. 12, 2021, the Tufts Center for the Study of Drug Development (Tufts CSDD) reported that ever…

On Jan. 12, 2021, Moderna announced that Swissmedic, the Swiss Agency for Therapeutic Products, had authorized the COVID-19…

On Jan. 11, 2021, Roche announced that the European Commission (EC) had approved Xofluza (baloxavir marboxil) for the…

On Jan. 11, 2021, Editas Medicine announced the U.S. Food and Drug Administration (FDA) had cleared the initiation…

On Jan. 11, 2021, Baxter announced an agreement to provide sterile manufacturing services for NVX-CoV2373, Novavax COVID-19 recombinant…
On Jan. 11, 2021, T2 Biosystems announced that its T2SARS-CoV-2 Panel – a molecular diagnostic test that detects…
On Jan. 11, 2021, Moderna announced that it was expanding its pipeline of innovative vaccines with three new…
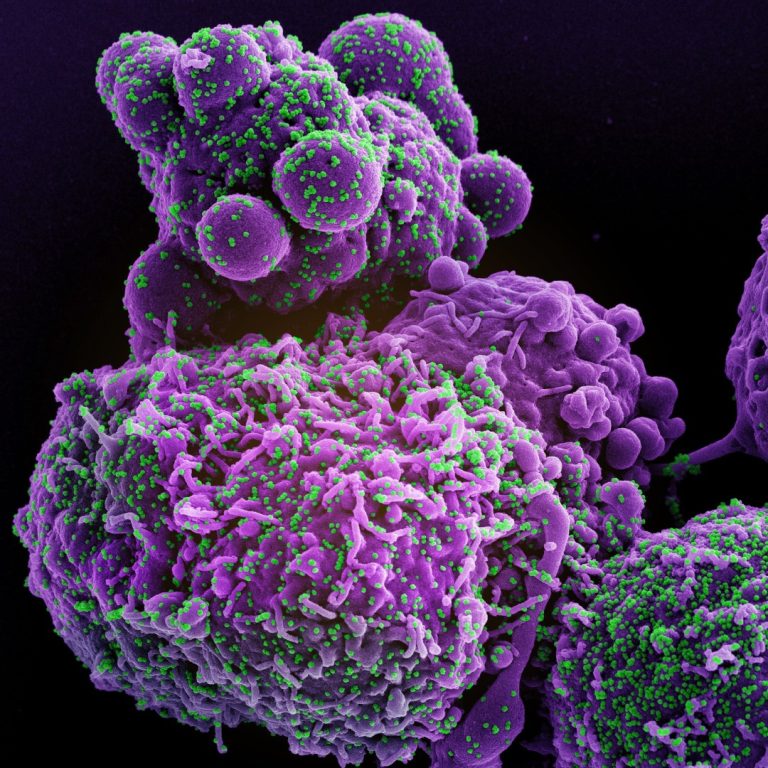
On Jan. 11, 2021, AXIM Biotechnologies announced that it had released a preprint of its manuscript describing the…

On Jan. 8, 2021, the American Cancer Society reported that the death rate from cancer in the U.S….

On Jan. 8, 2021, the COVID-19 vaccine developed by Moderna has today been given regulatory approval for supply…

On Jan. 8, 2021, the Medicines and Healthcare products Regulatory Agency (MHRA) announced it had accepted the recommendation…

On Jan. 8, 2021, Pfizer and BioNTech announced results from an in vitro study conducted by Pfizer and…

On Jan. 8, 2021, BioNTech announced that it was in talks with the European Commission (EC) about an…

On Jan. 7, 2021, Twist Bioscience announced it would supply the U.S. Centers for Disease Control and Prevention…