Luminex provided updates on critical efforts related to novel Coronavirus
On Mar. 4, 2020, Luminex announced they were working on multiple solutions to augment their NxTAG and ARIES…
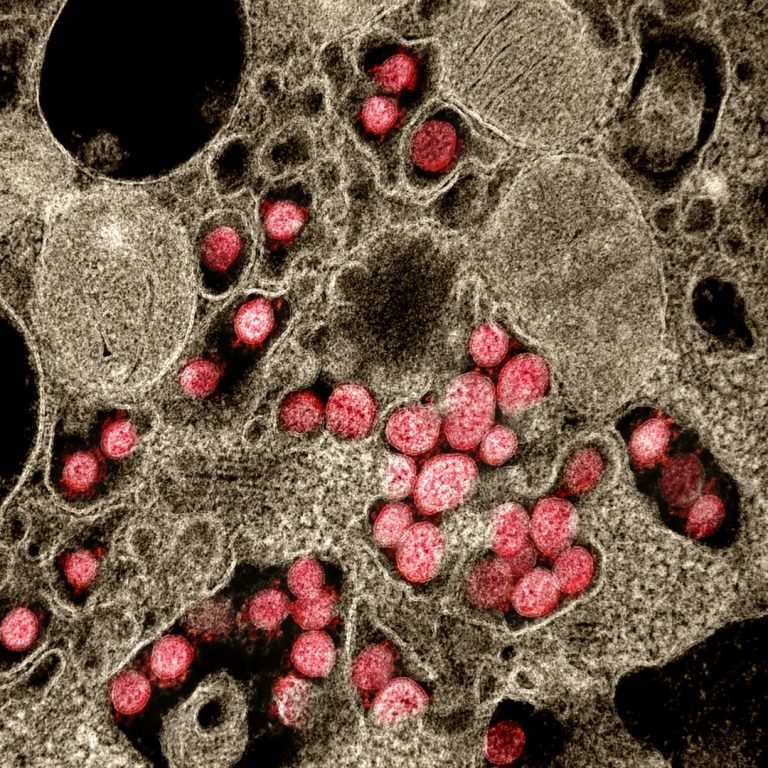
On Mar. 4, 2020, Luminex announced they were working on multiple solutions to augment their NxTAG and ARIES…

On Mar. 4, 2020, the UW Medicine Clinical Virology Lab announced it had received U.S. Food and Drug…
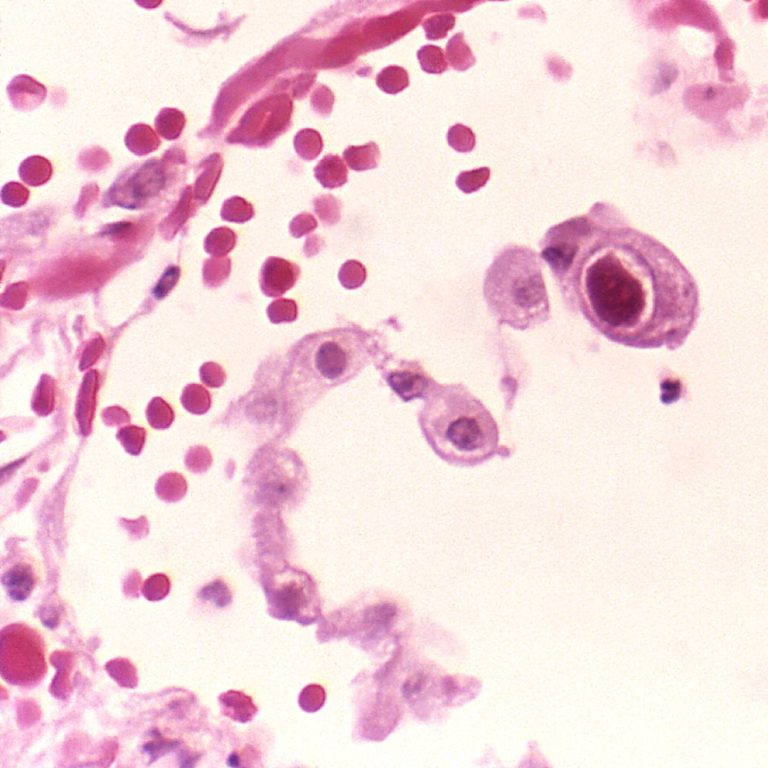
On Mar. 3, 2020, Moderna announced that enrollment was complete for all three dose cohorts of the Phase…

On Mar. 3, 2020, Heat Biologics announced the Company has formally launched a program within its wholly-owned subsidiary,…
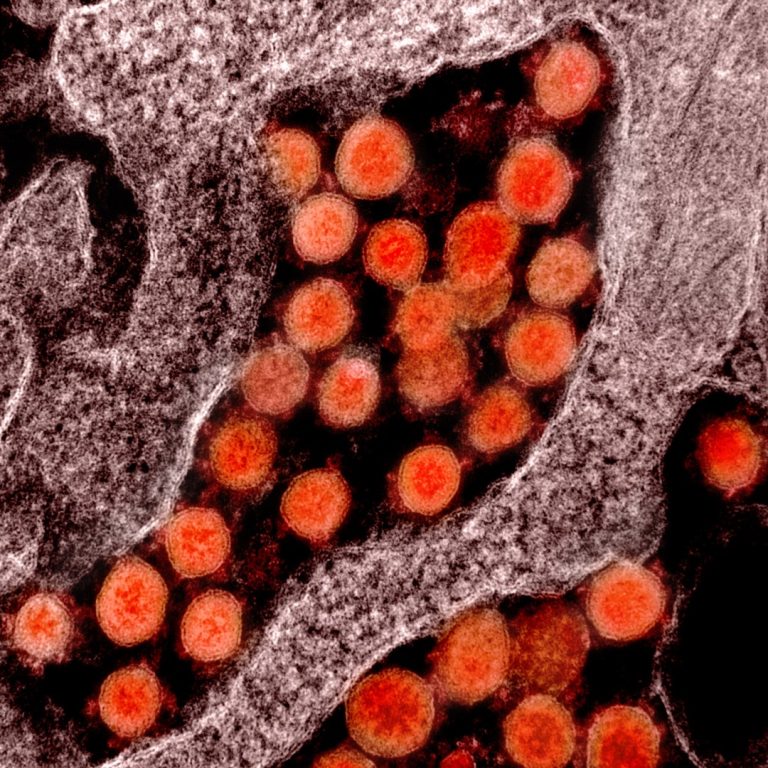
On Mar. 2, 2020, Boston’s top infectious disease researchers, including scientists from Boston University’s National Emerging Infectious Diseases…

On Mar. 2, 2020, Gilead Sciences announced it had acquired Forty Seven for $95.50 per share in cash…
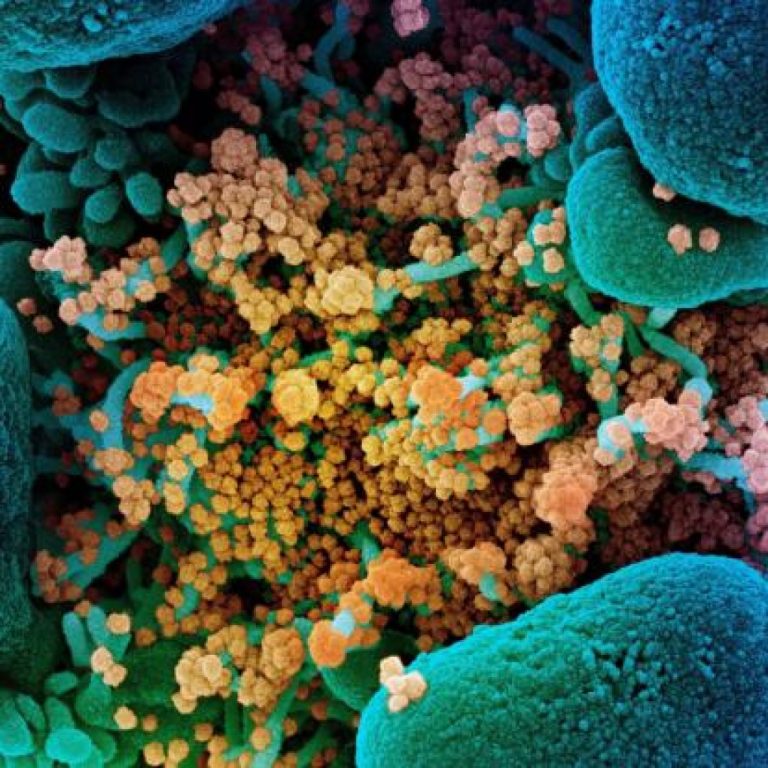
On Mar. 2, 2020, Mateon Therapeutics reported the company had been evaluating its therapeutic and AI platforms to…

On Mar. 2, 2020, Ophirex, a public-benefit biotechnology company working to improve outcomes for global victims of snakebite,…

On Mar. 2, 2020, Innovation Pharma provided the scientific rationale and clinical development perspectives for Brilacidin, the Company’s…

On Mar. 1, 2020, the United Nations Humanitarian Chief Mark Lowcock released US$15 million from the Central Emergency…

On Feb. 29, 2020, the University of Washington (UW) Medicine Clinical Virology Lab announced it had received U.S….

On Feb. 28, 2020, immediately after Christian Drosten published a genetic sequence of the novel coronavirus online, he…

On Feb. 27, 2020, Innovation Pharma announced the Company had signed a Material Transfer Agreement (MTA) with one…
On Feb. 27, 2020, a consortium of philanthropic, non-profit and private sector organisations launched a collaboration that aims…
On Feb. 25, 2020, the U. S. National Institutes of Health (NIH) announced it had launched a $1…

On Feb. 26, 2020, Georgia Institute of Technology and Shriners Hospitals for Children announced a collaborative research affiliation…

On Feb. 26, 2020, Novavax announced progress in its efforts to develop a novel vaccine to protect against…
On Feb. 26, 2020, Tonix Pharmaceuticals announced a strategic collaboration with Southern Research to support the development of…
On Feb. 25, 2020, the National Institutes of Health (NIH) announced that a randomized, controlled clinical trial to…

On Feb. 25, 2020, the Cherokee Nation became the first U.S. tribe to preserve culturally important seeds in…
On Feb. 25, 2020, a University of British Columbia (UBC) researcher was part of an international team working…
On Feb. 25, 2020, Vir Biotech and WuXi Biologics announced a development and manufacturing collaboration to advance and…

On Feb. 24, 2020, researchers at National Institute of Allergy and Infectious Disease’s Rocky Mountain Laboratories in Hamilton,…

On Feb. 24, 2020, Moderna announced it had released the first batch of mRNA-1273, the Company’s vaccine against…

On Feb. 24, 2020, Seqirus announced that the U.S. Food and Drug Administration (FDA) had approved the first…

On Feb. 21, 2020, Lundbeck announced that VYEPTI™ (eptinezumab-jjmr) had been approved by the U.S. Food and Drug…

On Feb. 21, 2020, a $2 million gift from University of Missouri alumni will support the university’s NextGen…

On Feb. 21, 2020, the U.S. Food and Drug Administration (FDA) authorized marketing of the first test to…
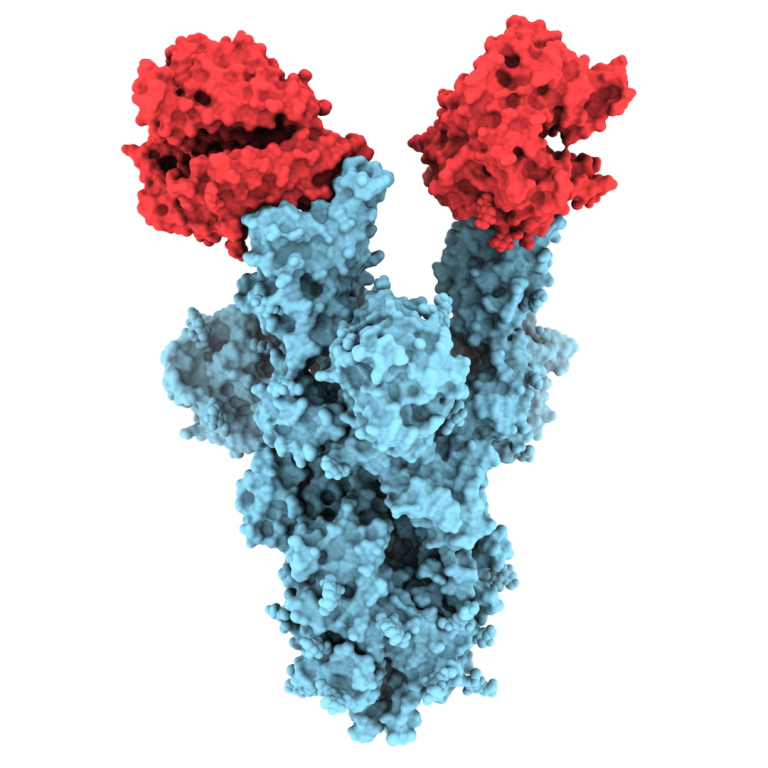
On Feb. 24, 2020, researchers reported that the spikes crowning the new coronavirus that causes COVID-19 atypical pneumonia…

On Feb. 21, 2020, the U.S. Food and Drug Administration (FDA) approved NEXLETOL (bempedoic acid) tablet, an oral,…