U.S. spending $5 billion to speed up development of new COVID vaccines
On Apr. 10, 2023, the U.S. Department of Health and Human Services (HHS) announced it was spending over…
On Apr. 10, 2023, the U.S. Department of Health and Human Services (HHS) announced it was spending over…
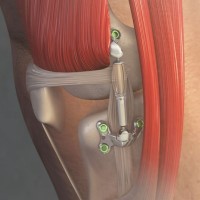
On Apr. 10, 2023, the U.S. Food and Drug Administration authorized for marketing Moximed’s MISHA Knee System, an…

On Apr. 7, 2023, the U.S. National Park Service reported Highly Pathogenic Avian Influenza (HPAI) had been confirmed…

On Apr. 5, 2023, researchers at Oregon Health & Science University’s OHSU Casey Eye Institute announced that gene…

On Apr. 5, 2023, Pfizer announced that experimental respiratory syncytial virus (RSV) vaccine was 82% effective in preventing…

On Apr. 5, 2023, Aberdeen Proving Ground South, formerly known as Edgewood, celebrated the opening of the Department…

On Apr. 5, 2023, the Food and Agriculture Organization (FAO), in collaboration with the World Health Organization (WHO),…
On Apr. 4, 2023, National Institutes of Health (NIH) funded research was announced from the University of Rochester…

On Apr. 4, 2023, NanoViricides announced that it had executed a license agreement with Karveer Meditech, Kolhapur, India,…

On Apr. 3, 2023, a study from the National Eye Institute (NEI) identified rare genetic variants that could…

On Apr. 3, 2023, the U.S. Department of Health and Human Services (HHS) released a National Cancer Plan,…

On Mar. 31, 2023, the Oregon Department of Agriculture and the United States Department of Agriculture’s Animal and…

On Mar. 30, 2023, Moderna and the Government of the Republic of Kenya announced they had finalized an…

On Mar. 29, 2023, the mortality gap between the United States and other high-income nations substantially expanded during…

On Mar. 29, 2023, the U.S. Food and Drug Administration (FDA) announced approval of Narcan, 4 milligram naloxone…

On Mar. 29, 2023, the World Health Organization (WHO) announced that it had certified Azerbaijan and Tajikistan for…

On Mar. 28, 2023, the American Chemical Society awarded The Priestley Medal to Cato T. Laurencin “to recognize distinguished…

On Mar. 27. 2023, Thermo Fisher Scientific and the University of California, San Francisco, announced they will accelerate…

On Mar. 27, 2023, CIRM Board approved investing nearly $26 million in 17 projects in early-stage discovery research.The…

On Mar. 26,. 2023, Guangdong Centers for Disease Control in China reported an H3N8 infection that would become…
On Mar. 24, 2023, the U.S. Centers for Disease Control and Prevention (CDC) announced that Tuberculosis (TB) in…

On Mar. 24. 2023, antibiotics do not reduce the risk of dying in adults hospitalized with COVID-19, the…

On Mar. 23. 2023, researchers at Washington University School of Medicine in St. Louis and the Veterans Affairs…
On Mar. 23, 2023, the Fred Hutchinson Cancer Center (FHCRC) announced a new building in South Lake Union…

On Mar. 23, 2023, a study published in Scientific Reports warned that vibrio vulnificus is an increasingly recognized…

On Mar. 23. 2023, scientists at the National Research Institute for Agriculture, Food and the Environment in Paris…

On Mar. 22, 2023, the White House Office of Science and Technology Policy released a series of five…

On Mar. 22. 2023, scientists at the University of Cambridge announced that Beethovenメs genome had been sequenced for…

On Mar. 21, 2023, the Ministry of Health of the United Republic of Tanzania declared an outbreak of…

On Mar. 21, 2023, Quest Diagnostics announced that it had introduced two innovative Post-COVID-19 panels. These laboratory tests…