CytoDyn submited protocol with FDA for phase 3 Registrational trial of leronlimab for critically ill COVID-19 population
On Dec. 9, 2021, CytoDyn announced that it had submitted a Phase 3, randomized, double blind, placebo controlled…

On Dec. 9, 2021, CytoDyn announced that it had submitted a Phase 3, randomized, double blind, placebo controlled…

On Dec. 9, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had expanded…

On Dec. 8, 2021, PerkinElmer announced the research use only launch of the NEXTFLEXᆴ Variant-Seqル SARS-CoV-2 Kit v2…

On Dec. 8, 2021, the U.S. Food and Drug Administration issued an emergency use authorization for AstraZenecaメs Evusheld…

On Dec. 8, 2021, Pfizer and BioNTech announced results from an initial laboratory study demonstrating that serum antibodies…

On Dec. 8, 2021, in a large-scale study of people from diverse ancestries, researchers narrowed down the number…

On Dec. 7, 2021, Rockefeller University scientists announced a study had demonstrated the therapeutic potential of an unusual…

On Dec. 7, 2021, Arbutus Biopharma, X-Chem, and Proteros announced that Arbutus had identified several molecules that inhibit…

On Dec. 7, 2021, the Centers for Disease Control and Prevention announced that it had awarded $22 million…

On Dec. 6, 2021, Roche announced that the European Medicines Agencyメs Committee for Medicinal Products for Human Use…

On Dec. 6, 2021, the World Health Organization Guideline Development Group of international experts announced in the BMJ…

On Dec. 6, 2021, the USAID announced the foundation of a new whole-of-government effort, the Initiative for Global…

On Dec. 5, 2021, Sorrento Therapeutics announced the peer-reviewed publication of a series of novel SARS-CoV-2 MPro inhibitors…

On Dec. 5, 2021, Roche announced that its planned to launch the SARS-CoV-2 & Flu A/B Rapid Antigen…

On Dec. 5, 2021, Johnson & Johnson announced preliminary results from an independent study, including a subset of…

On Dec. 3, 2021, Roche and TIB Molbiol announced they had added three additional Research Use Only test…

On Dec. 3, 2021, Gavi, the Vaccine Alliance, approved a new malaria vaccination programme to support the introduction,…

On Dec. 3, 2021, Genomics England has announced its vision for a pilot research project to examine the…

On Dec. 3, 2021, a report from the U.S. Department of Health and Human Services (HHS) found that…

On Dec. 2, 2021, researchers at the National Eye Institute (NEI) announced they had identified, isolated, and characterized…

On Dec. 2, 2021, National Resilience and Harvard University announed they had established a five-year research and development…

On Dec. 1, 2021, Moderna announced a revised supply agreement with the UK government for up to 60…
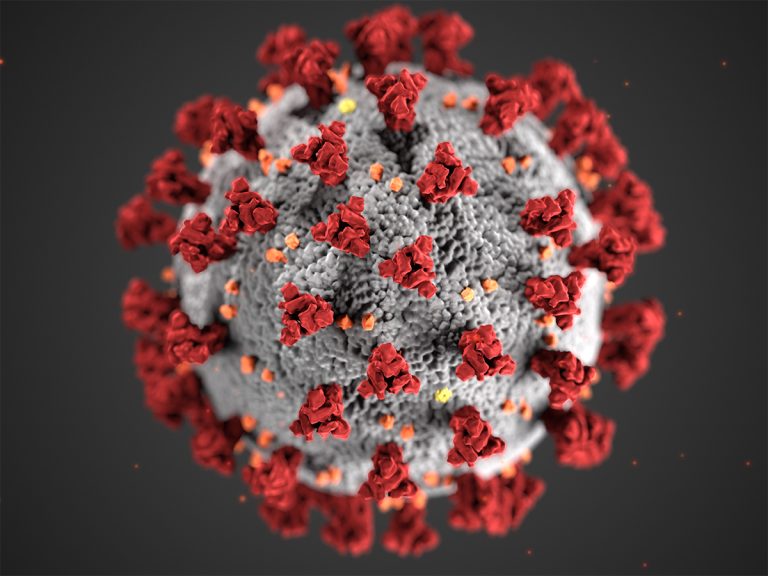
On Dec. 1, 2021, the California and San Francisco Departments of Public Health confirmed that a recent case…

On Dec. 1, 2021, Fulgent Genetics confirmed that the Companyメs RT-PCR test for SARS-CoV-2, the virus that causes…
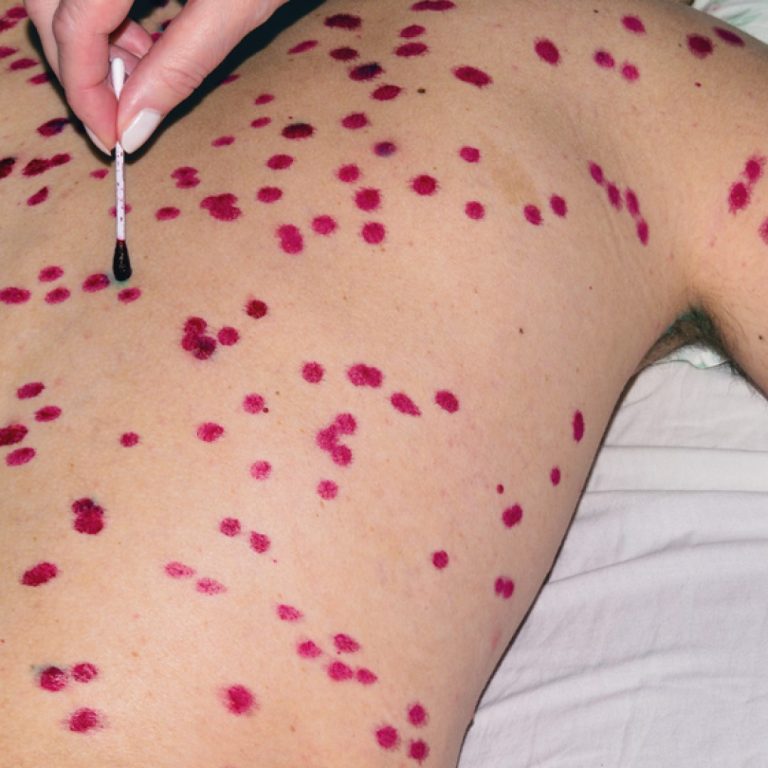
On Dec. 1, 2021, SIGA Technologies announced that Health Canada had approved oral TPOXX (tecovirimat) as an extraordinary…
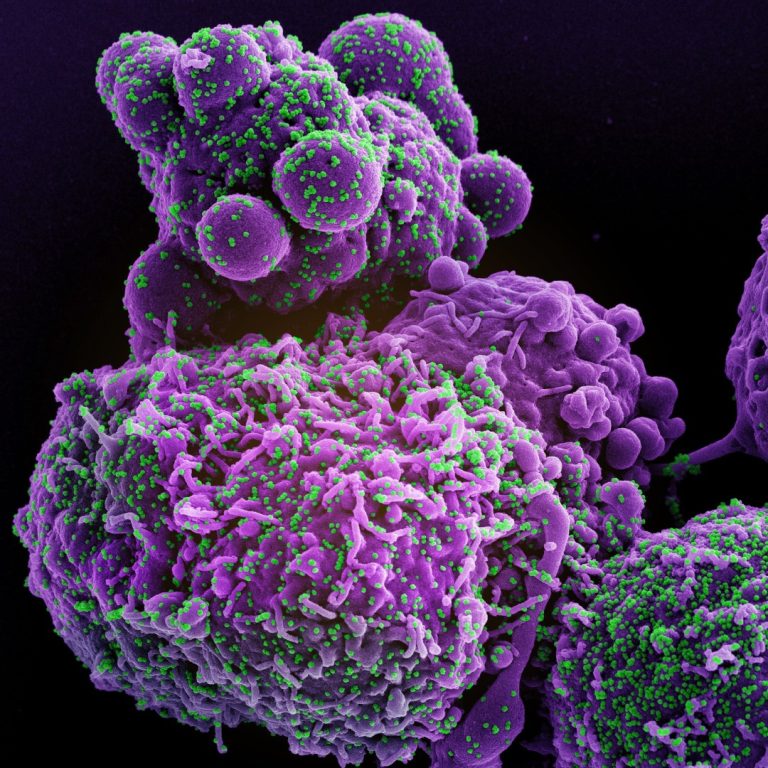
On Dec. 1, 2021, T2 Biosystems announced that its T2SARS-CoV-2 Panel detected the Omicron COVID-19 variant (B.1.1.529). The…
On Nov. 30, 2021, LabCorp announced that it had begun offering observed self-collection for COVID-19 PCR testing in…

On Nov. 29, 2021, Thermo Fisher Scientific confirmed that its polymerase chain reaction (PCR) TaqPath COVID-19 Combo Kit…
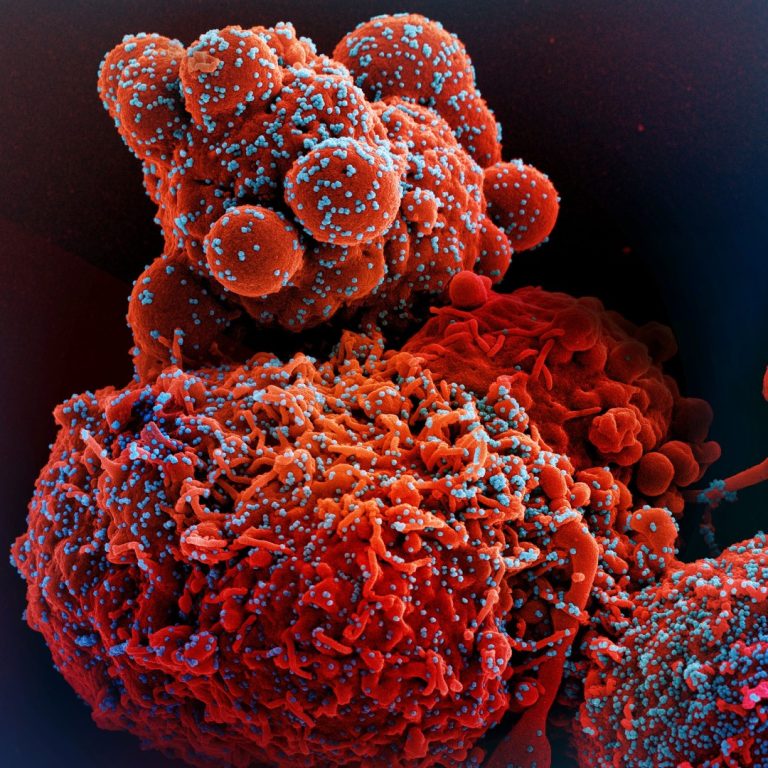
On Nov. 29, 2021, Hologic announced that its three SARS-CoV-2 tests all detect the recently emerged Omicron variant of…
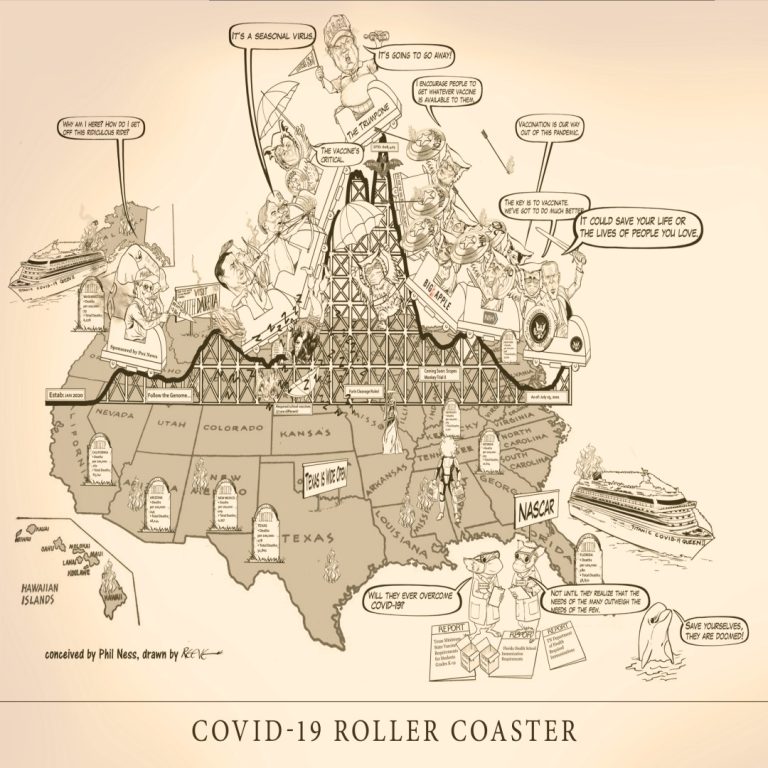
The COVID-19 Roller Coaster is a wild ride, strap yourself in and hold on… Cast of Characters: Senator…