FDA approved two emergency INDs for COVID-19 outpatient therapy
On Aug. 6, 2020, Organicell Regenerative Medicine announced that the FDA had approved two outpatient Emergency Investigational New…

On Aug. 6, 2020, Organicell Regenerative Medicine announced that the FDA had approved two outpatient Emergency Investigational New…

On Aug. 6, 2020, the world’s first successfully cloned endangered Przewalski’s horse was born. Revive & Restore, San…

On Aug. 6, 2020, researchers at the AIDS Institute, Department of Microbiology and State Key Laboratory of Emerging…

On Aug. 5, 2020, the Tecan Group and Thermo Fisher Scientific announced a collaboration to enable scaled-up COVID-19…

On Aug. 5, 2020, BioNTech and Shanghai Fosun Pharmaceutical announced that the first 72 participants had been dosed…

On Aug. 5, 2020,Thermo Fisher Scientific announced a new highly automated, real-time PCR solution designed to analyze up…

On Aug. 5, 2020, Johnson & Johnson announced a $1 billion agreement with the U.S. government for the…

On Aug. 5, 2020, Pfizer Canada and BioNTech announced an agreement with the Government of Canada to supply…

On Aug. 5, 2020, the Cancer Research Institute (CRI), a U.S. nonprofit organization dedicated to the discovery and…

On Aug. 5, 2020, Variation Biotechnologies, an Ottawa-based subsidiary of VBI Vaccines, announced that it had been awarded…

On Aug. 5, 2020, Pfizer and Shanghai Fosun Pharmaceutica announced that the first 72 participants had been dosed…
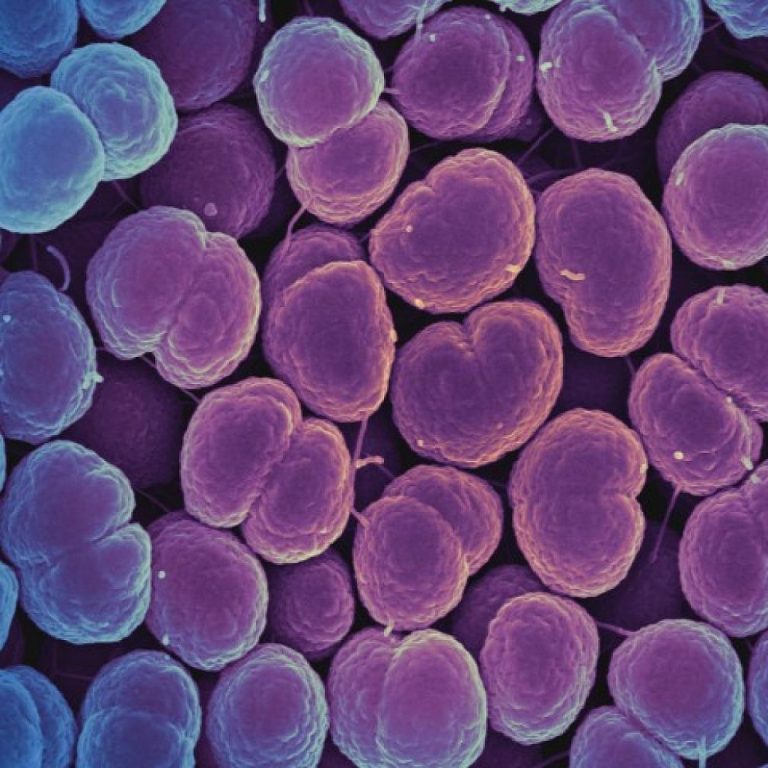
On Aug. 5, 2020, a diagnostic test capable of accurately and reliably detecting the microorganism that causes gonorrhea…

On Aug. 5, 2020, a randomized, controlled clinical trial evaluating the safety and efficacy of a treatment regimen…

On Aug. 5, 2020, the National Institutes of Health (NIH) announced it had launched the Medical Imaging and…

On Aug. 5, 2020, the National Institute of Allergy and Infectious Diseases (NIAID) announced that the investigational vaccine…

On Aug. 5, 2020, Heat Biologics highlighted the importance of T cell immunity in preventing COVID-19 in a…

On Aug. 5, 2020, AXIM Biotechnologies announced the development, patent filing and Emergency Use Approval (EUA) filing of…

On Aug. 4, 2020, Innovation Pharma reported receiving an update from a U.S. Regional Biocontainment Laboratory (RBL) based…

On Aug. 4, 2020, Abiomed announced the FDA had issued an emergency use authorization (EUA) for its left-sided…
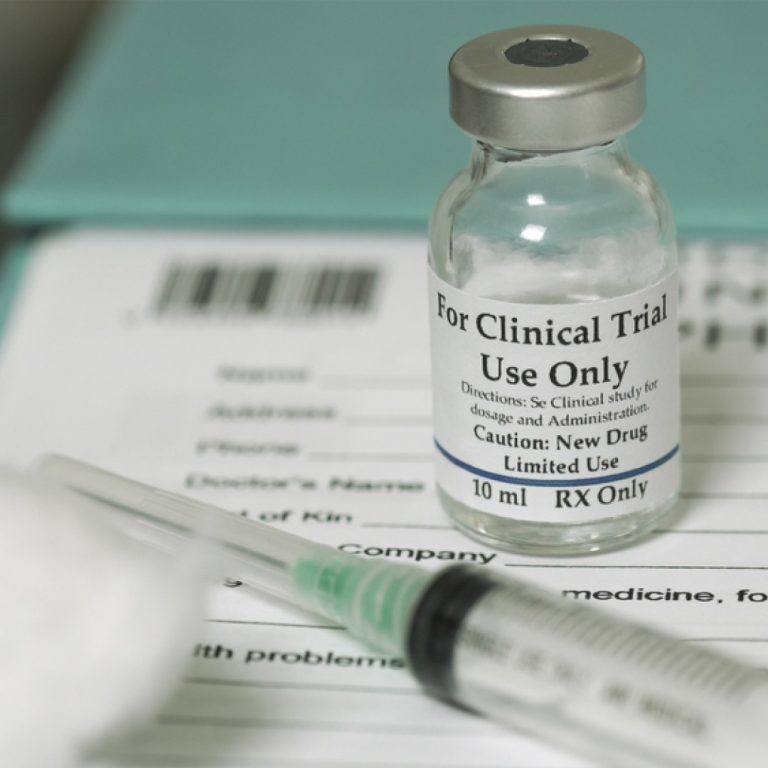
On Aug. 4, 2020, Novavax announced Phase 1 data from its Phase 1/2 randomized, observer-blinded, placebo-controlled trial of…

On Aug. 4, 2020, Cocrystal Pharma announced the publication of preclinical animal studies of coronavirus antiviral compounds in…

On Aug. 4, 2020, researchers at The University of Hong Kong (HKUMed) collaborated with researchers at the Institute…

On Aug. 4, 2020, Yale University announced it was among the core partners of the new Center for…

On Aug. 4, 2020, Mateon Therapeutics and Abiogenesis announced they had initiated a ARTI-19 randomized, controlled, multi-site India…

On Aug. 4, 2020, Precipio announced it had completed the internal validation of the U.S. Food and Drug…

On Aug. 4, 2020, The Centers for Disease Control and Prevention (CDC) anticipates that 2020 will be another…

On Aug. 4, 2020, the NIH announced that patients admitted with COVID-19 at select hospitals may now volunteer…

On Aug. 3, 2020, members of the COVID R&D Alliance AbbVie, Amgen and Takeda Pharma announced the first…
On Aug. 3, 2020, Todos Medical announced a partnership with PATHNOVA, a Singapore-based clinical laboratory, for the Company’s…

On Aug. 3, 2020, Atossa Therapeutics announced that it had received approval from the ethics committee to open…