RECOVERY trial closed recruitment to convalescent plasma treatment for patients hospitalised with COVID-19
On Jan. 15, 2021, Oxford University announced that on advice of the independent Data Monitoring Committee (DMC), recruitment…

On Jan. 15, 2021, Oxford University announced that on advice of the independent Data Monitoring Committee (DMC), recruitment…

On Jan. 15, 2021, VBI Vaccines announced that results from a Phase 4 study of VBIメs prophylactic 3-antigen…

On Jan. 15, 2021, scientists at Washington University School of Medicine in St. Louis announced that that a…

On Jan. 15, 2021, Pfizer and BioNTech announced they had developed a plan that allowed the scale-up of…

On Jan. 15, 2021, researchers at the University of Wisconsin School of Medicine and Public Health and UW…

On Jan. 14, 2021, Twist Bioscience announced that it had started shipping its new synthetic RNA reference controls,…

On Jan. 14, 2021, Innovation Pharmaceuticals announced that the FDA had designated as a Fast Track development program…

On Jan. 14, 2021, PerkinElmer announced that its PerkinElmerᆴ Coronavirus Nucleic Acid Detection Kit received Emergency Use Authorization…

On Jan. 14, 2021, researchers from the University of Southern California and Princeton reported that the COVID-19 pandemic,…

On Jan. 14, 2021, Mammoth Biosciences announced that it had secured a subcontract with MRIGlobal, the prime contractor…

On Jan. 14, 2021, Cepheid that Health Canada announced that it had issued Cepheid a medical device license…

On Jan. 13, 2021, researchers announced they had developed a blood test that could make it possible for…

On Jan. 13, 2021, National Institutes of Health scientists and their colleagues announced they had identified a S….

On Jan. 13, 2021, Oxford Immunotec announced the start of a collaboration with Valneva whereby it will perform…
On Jan. 13, 2021, Mateon Therapeutics reported positive interim results from its ARTI-19 clinical trial evaluating ARTIVedaTM against…

On Jan. 13, 2021, the USDAメs (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 (the virus that…

On Jan. 13, 2021, Alexion announced the decision to pause further enrollment in the global Phase 3 study…

On Jan. 13, 2021, SIGA Technologies announced that the Public Health Agency of Canada had awarded a contract…

On Jan. 13, 2021, the Oregon Department of Agriculture (ODA) continueed to test, survey, and trap at an…

On Jan. 12, 2021, the four leading international health and humanitarian organizations announced the establishment of a global…

On Jan. 12, 2021, Merck announced the FDA accepted for priority review a Biologics License Application (BLA) for…

On Jan. 12, 2021, Abbott announced it had received FDA 510(k) clearance for the first rapid handheld traumatic…

On Jan. 12, 2021, the WHO announced that global scientists were intensifying research into COVID-19 to expand its…

On Jan. 12, 2021, UC Berkeley HIV researchers announced that they had determined the atomic structure of a…

On Jan. 12, 2021, the Tufts Center for the Study of Drug Development (Tufts CSDD) reported that ever…

On Jan. 12, 2021, Moderna announced that Swissmedic, the Swiss Agency for Therapeutic Products, had authorized the COVID-19…

On Jan. 12, 2021, Regeneron announced that the U.S. Department of Health and Human Services (HHS) and the…
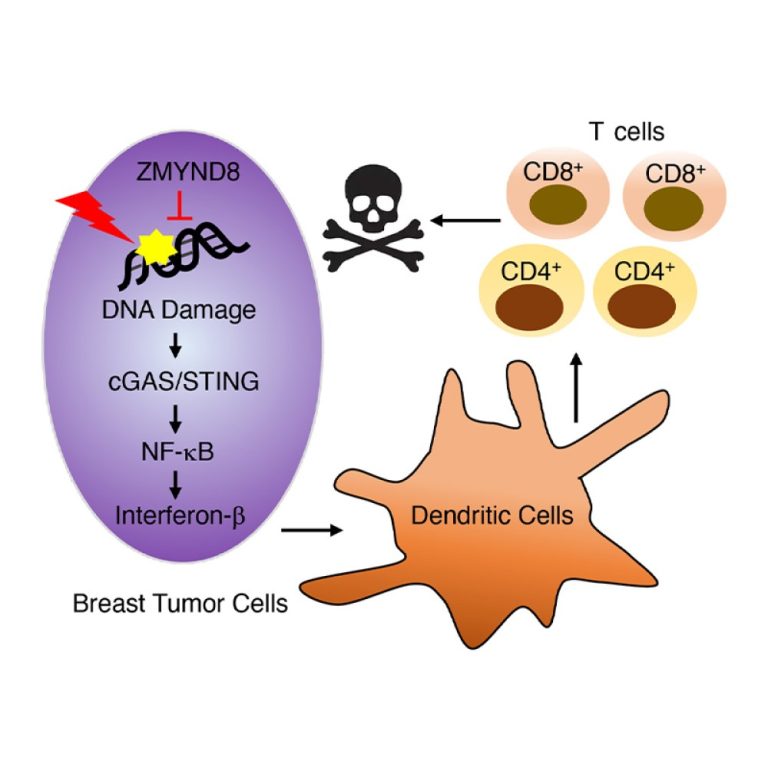
On Jan. 12, 2021, a team researchers from the University of Texas Southwestern (UTSW) announced they had identified…

On Jan. 11, 2021, the FDA conditionally approved Anivive Lifesciences’ Laverdia-CA1 (verdinexor tablets) to treat dogs with lymphoma,…

On Jan. 11, 2021, Editas Medicine announced the U.S. Food and Drug Administration (FDA) had cleared the initiation…