Roche announced FDA Emergency Use Authorization and availablabilityof cobas SARS-CoV-2 detection test
On Mar. 13, 2020, Roche announced the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization…

On Mar. 13, 2020, Roche announced the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization…

On Mar. 13, 2020, Thermo Fisher Scientific announced the U.S. Food and Drug Administration (FDA) has issued an emergency…

On Mar. 13, 2020, declared a COVID-related emergency under Section 501(b) of the Stafford Act [18]. Under the…

On Mar. 13, 2020, Washington University in St. Louis announced the first long-term study of Bacillus thuringiensis (Bt)…
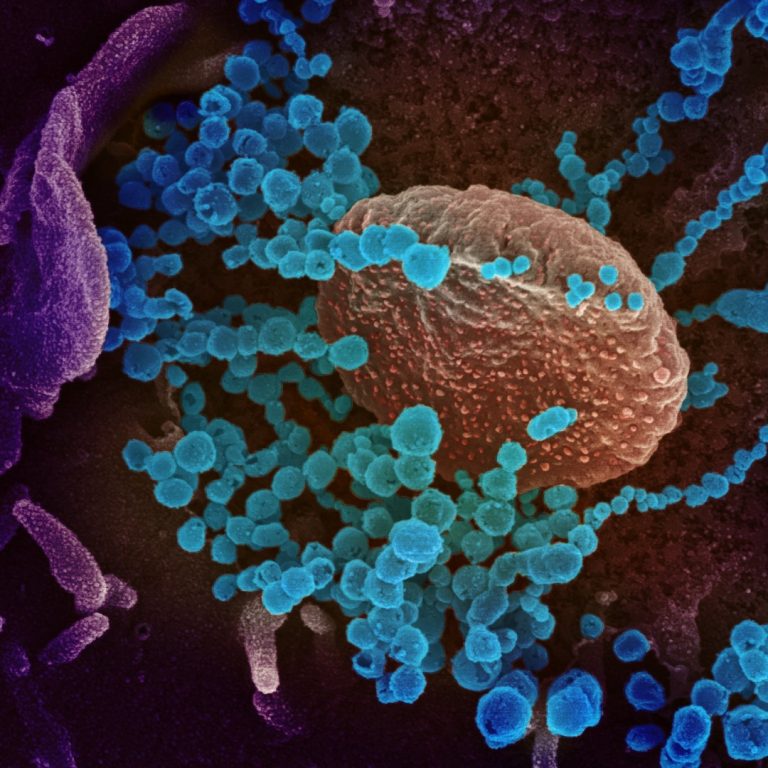
On Mar. 13, 2020, Enanta Pharmaceuticals announced it had initiated a program to discover direct-acting antiviral drug candidates…

On Mar. 13, 2020, IDEXX Laboratories announced the company had seen no positive results in pets to date…

On Mar. 13, 2020, Johnson & Johnson announced that its Janssen Pharmaceutical Companies have entered a collaboration with…

On Mar. 13, 2020, BioReference Laboratories, an OPKO company, announced it had begun accepting specimens for testing of…

On Mar. 13, 2020, Luminex announced that four independent clinical laboratories had validated laboratory developed tests (LDTs) for…

On Mar. 13, 2020, PATH and the Centers for Disease Control and Prevention (CDC) announced a collaboration with…

On Mar. 13, 2020, researchers at the National Eye Institute (NEI) reported finding a protein that normally deposits…
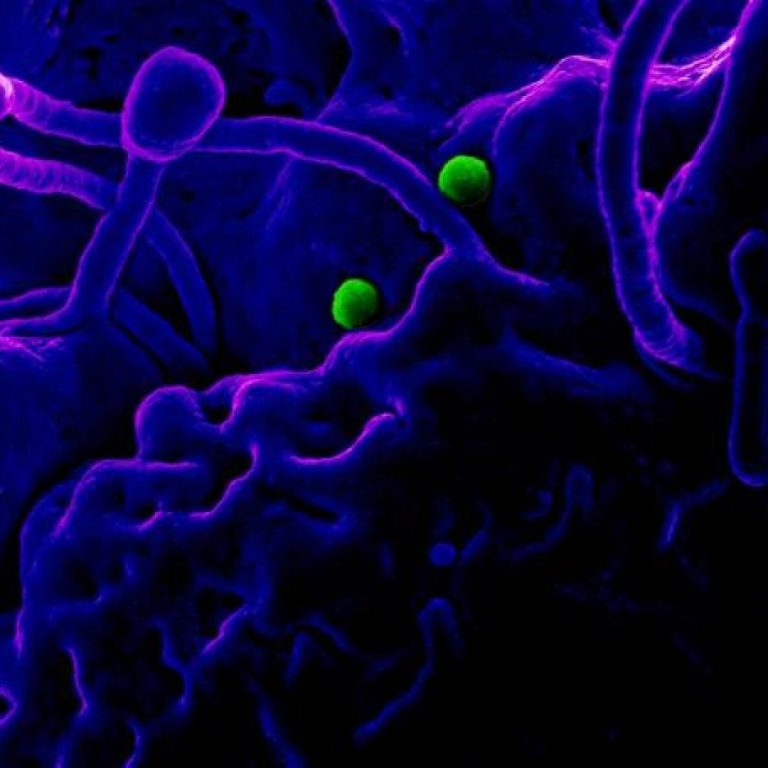
On May 12, 2022, the National IHR Focal Point for Qatar reported two laboratory-confirmed cases of Middle East…

On Mar. 12, 2020, Pluristem Therapeutics announced it a collaborative agreement with the BIH Center for Regenerative Therapy…
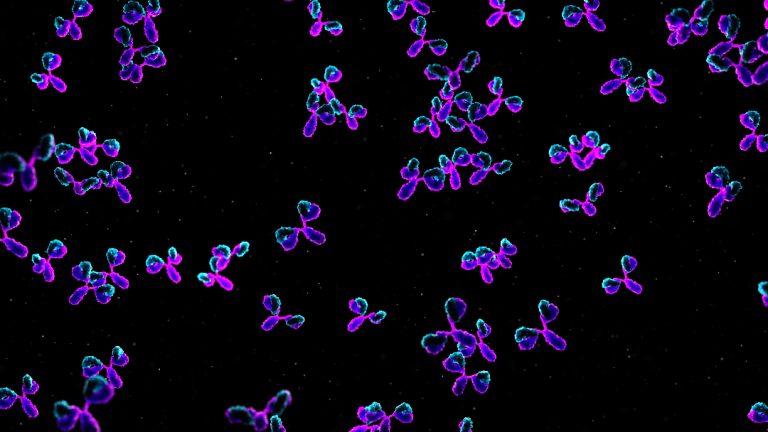
On Mar. 12, 2020, Vir Biotech announced it had signed a letter of intent with Biogen for the…

On Mar. 12, 2020, Emory University announced it was participating in a National Institutes of Health (NIH) sponsored…

On Mar. 12, 2020, AbCellera and Eli Lilly announced an agreement to co-develop antibody products for the treatment…

On Mar. 12, 2020, Innovation Pharma announced research procedures that one of the 12 Regional Biocontainment Labs (RBLs)…

On Mar. 12, 2020, Predictive Oncology announced it will launch a new AI platform for vaccine and drug…
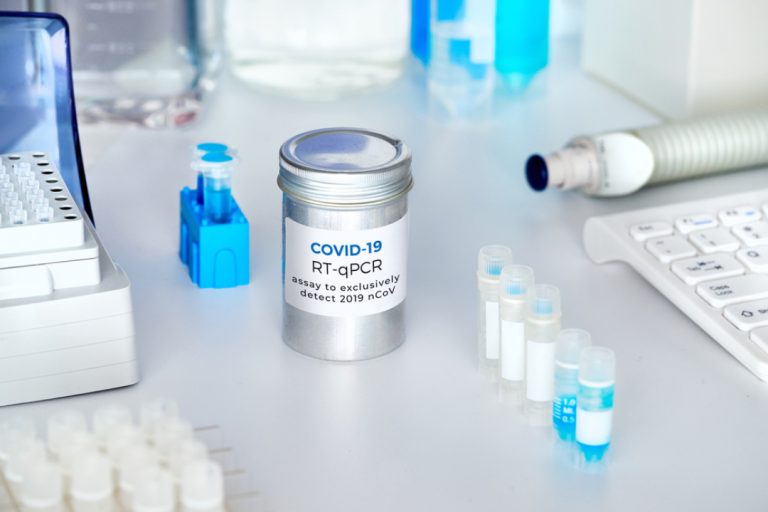
On Mar. 12, 2020, the Mayo Clinic announced it had developed a test that can detect the SARS-CoV-2…

On Mar. 11 2020, the World Health Organization declared the Coronavirus outbreak a global pandemic. To-date, of the…

On Mar. 11, 2020, the antiretroviral drugs dolutegravir and emtricitabine/tenofovir alafenamide fumarate (DTG+FTC/TAF) may comprise the safest and…

On Mar. 11, 2020, the Howard Hughes Medical Institute (HHMI) announced a new investment of over $120 million…
On Mar. 11, 2020, bioMerieux announced the forthcoming launch of 3 different tests to address the COVID-19 epidemic…

On Mar. 11, 2020, Vir Biotech announced a research collaboration agreement with the NIH to advance characterization and…

On Mar. 11, 2020, the La Jolla Institute for Immunology (LJI) and the J. Craig Venter Institute announced…

On Mar. 11, 2020, Emergent BioSolutions announced it has initiated development of two product candidates for treatment and…

On Mar. 10, 2020, the Bill & Melinda Gates Foundation, Wellcome, and Mastercard committed up to $125 million…

On Mar. 10, 2020, CaroGen and scientists from Yale announced they had begun work on a vaccine targeting…

On Mar. 10, 2020, Innovation Pharma announced that management was advised that testing of Brilacidin, the Company’s flagship…

On Mar. 10, 2020, BCerTest Biotec, along with BD (Becton, Dickinson and Company) announced that the VIASURE SARS-CoV-2…