FDA approved prosthetic implant for above-the-knee amputations
On Dec. 18, 2020, the FDA approved the Osseoanchored Prostheses for the Rehabilitation of Amputees (OPRA) Implant System,…

On Dec. 18, 2020, the FDA approved the Osseoanchored Prostheses for the Rehabilitation of Amputees (OPRA) Implant System,…

On Dec. 18, 2020, the National Research Council of Canada (NRC) announced it was providing advisory services and…

On Dec. 18, 2020, Johnson & Johnson announced that its subsidiary Janssen Pharma, will provide up to 500…

On Dec. 18, 2020, the World Health Organization (WHO) announced that it had arrangements in place to access…

On Dec. 18, 2020, the the U.S. Food and Drug Administration (FDA) approved AstraZeneca’s Tagrisso (osimertinib) as the…

On Dec. 18, 2020, Moderna announced that the Centers for Disease Control and Prevention’s (CDC) Advisory Committee on…

On Dec. 18, 2020, Eli Lilly announced plans to begin a new pragmatic study of bamlanivimab (LY-CoV555) in…

On Dec. 18, 2020, Moderna announced that the U.S. Food and Drug Administration (FDA) had authorized the emergency…

On Dec. 17, 2020, PRA Health Sciences announced the enhancement of its COVID-19 Monitoring Program with the addition…

On Dec. 17, 2020, XPhyto Therapeutics and 3a-diagnostics announced the successful validation of their point-of-care SARS-CoV-2 (COVID-19) RT-PCR…

On Dec. 17, 2020, Regeneron announced that the New England Journal of Medicine (NEJM) had published initial clinical…
On Dec. 17, 2020, JAMA reported that the daily U.S. mortality rate for COVID-19 deaths was equivalent to…

On Dec. 17, 2020, Johnson & Johnson announced that the large-scale, pivotal, multi-country Phase 3 trial (ENSEMBLE) of…

On Dec. 17, 2020, NIAID announced that an observational study had launched to evaluate the short- and long-term…
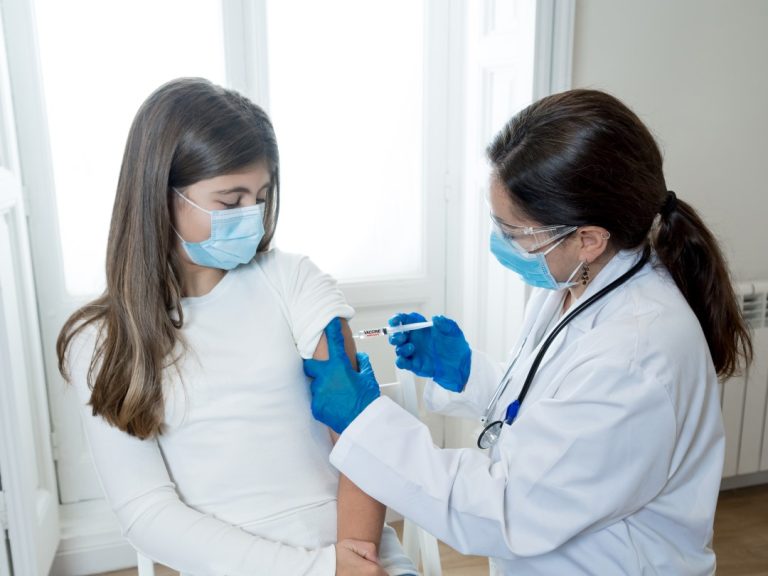
On Dec. 17, 2020, Moderna confirmed that the U.S. Food and Drug Administration’s (FDA) Vaccines and Related Biological…
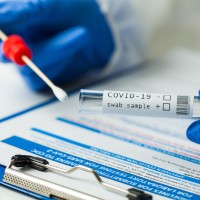
On Dec. 17, 2020, ACON Laboratories announced that its SARS-COV-2 IgG/IgM Rapid Test has been authorized for emergency…

On Dec. 17, 2020, NIAID announced that two randomized, controlled Phase 3 clinical trials had begun evaluating investigational…

On Dec. 17, 2020, Quest Diagnostics and The Commons Project, a non-profit public trust dedicated to building and…

On Dec. 16, 2020, Novavax announced an Advance Purchase Agreement with the government of New Zealand for the…

On Dec. 16, 2020, Heat Biologics announced it had completed its gp96-based COVID-19 vaccine cell line, which is…

On Dec. 16, 2020, BD (Becton, Dickinson) announced that it had received pandemic orders for needles and syringes…

On Dec. 16, 2020, Pacific Biosciences announced initial findings from the companyメs research collaboration with Labcorp that was…

On Dec. 16, 2020, the U.S. Food and Drug Administration issued a new emergency use authorization (EUA) for…

On Dec. 16, 2020, Abbott announced that the FDA had issued Emergency Use Authorization (EUA) for virtually guided…

On Dec. 16, 2020, Meridian Bioscience announced that it had been awarded funding in the amount of approximately…

On Dec. 16, 2020, BioNTech and Shanghai Fosun Pharmaceutical announced an agreement to supply Mainland China with an…

On Dec. 16, 2020, National Institute of Allergy and Infectious Diseases reported that an observational study had launched…
On Dec. 15, 2020, AXIM Biotechnologies announced the development and patent filing for an enzyme-linked immunosorbent assay (ELISA)-based…

On Dec. 15, 2020, Inovio Pharma announced the company and a team of scientists from The Wistar Institute,…
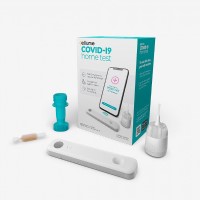
On Dec. 15, 2020, the FDA granted emergency use authorization (EUA) for the first over-the-counter (OTC) fully at-home…