CytoDyn reached enrollment of 195 patients in its phase 3 trial for COVID-19 patients with severe-to-critical symptoms
On Aug. 25, 2020, CytoDyn announced the Company had reached the requisite number of enrolled patients in its…
On Aug. 25, 2020, CytoDyn announced the Company had reached the requisite number of enrolled patients in its…

On Aug. 24, 2020, Hong Kong University researchers reported that a 33-year-old man in Hong Kong had been…

On Aug. 24 2020, NantKwest and ImmunityBio announced an agreement to jointly develop, manufacture, market, and commercialize therapeutics…

On Aug. 24, 2020, the combination of hydroxychloroquine and azithromycin has been linked to significant cardiovascular risks, including…
On Aug. 24, 2020, a published study led by the Universities of Oxford and Birmingham found that, compared…

On Aug. 24, 2020, Innovation Pharma reported receiving additional data from a U.S. Regional Biocontainment Laboratory (RBL) based…

On Aug. 24, 2020, humans are not the only species facing a potential threat from SARS-CoV-2, the novel…

On Aug. 24, 2020, Novavax announced that the first volunteers were enrolled in the Phase 2 portion of…

On Aug. 24, 2020, XBiotech announced that the U.S. Food and Drug Administration (FDA) issued an emergency use…

On Aug. 24, 2020,Trevena announced that Imperial College London (ICL) had initiated a proof-of-concept study for TRV027 in…

On Aug. 24, 2020, AXIM Biotechnologies announced that it had signed an exclusive limited licensing, manufacturing and distribution…

On Aug. 24, 2020, Catalent announced that it had contracted to provide drug substance manufacturing to AstraZeneca for…

On Aug. 24, 2020, the Quest Diagnostics Foundation announced a wide-ranging initiative to address and reduce health disparities…

On Aug. 23, 2020, the FDA issued an emergency use authorization (EUA) for investigational convalescent plasma for the…
On Aug. 23, 2020, the Mayo Clinic announced that with the FDA Emergency Use Authorization of convalescent plasma,…

On Aug. 23, 2020, the World Heath Organization (WHO) reported that 172 economies were engaged in discussions to…

On Aug. 21, 2020, research by the University of Minnesota and the University of Washington found that every…
On Aug. 21, 2020, the CDC published Immunization Practices Advisory Committee (ACIP) 2020-21 influenza vaccination recommendations. Routine annual…

On Aug. 21, 2020, JAMA reported that hospitalized patients with moderate COVID-19 randomized to a 5-day course of…

On Aug. 20, 2020, Oragenics and Avid Bioservices announced a process development and drug substance manufacturing agreement. Avid…

On Aug. 20, 2020, Sorrento Therapeutics and SmartPharm Therapeutics announced the signing of a merger agreement under which…

On Aug. 20, 2020, GT Biopharma and Cytovance announced a collaboration agreement whereby Cytovance will providing drug development…
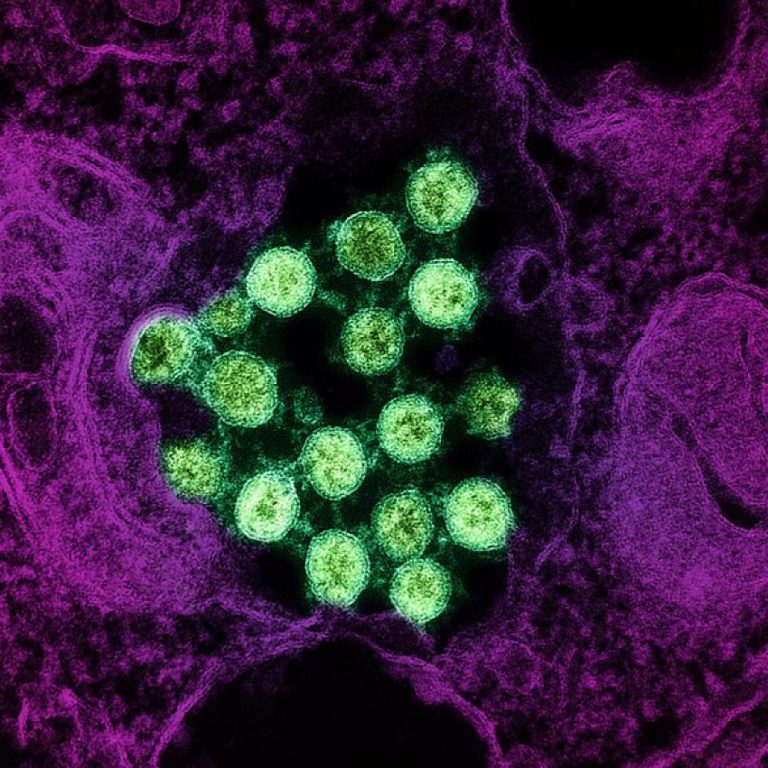
On Aug. 20, 2020, the National Institute of Allergy and Infectious Diseases (NIAID), one of the National Institutes…

On Aug. 20, 2020, in the most comprehensive study of COVID-19 pediatric patients to date, Harvard Medical School…

On Aug. 20, 2020, OpGen announced that its subsidiary Curetis GmbH had obtained the CE mark certification in…

On Aug. 20, 2020, Pfizer and BioNTech shared additional Phase 1 safety and immunogenicity data from their U.S….

On Aug. 19, 2018, BioNexus KC announced it had been awarded a $42,900 grant from the Health Forward…

On Aug. 19, 2020, Quidel announced that labeling for Quidel’s Emergency Use Authorization (EUA) for the Sofiaᆴ SARS…

On Aug. 19, 2020, Accelerate Diagnostics and BioCheck announced that the FDA had issued an Emergency Use Authorization…

On Aug. 19, 2020, the National Institutes of Health (NIH) began requiring a small nonprofit research organization to…