FDA amended EUA for Hologicメs Aptima SARS-CoV-2 assay to Include COVID-19 testing of asymptomatic individuals
On Oct. 6, 2020, Hologic announced that its Aptimaᆴ SARS-CoV-2 assay, which initially received Emergency Use Authorization (EUA)…

On Oct. 6, 2020, Hologic announced that its Aptimaᆴ SARS-CoV-2 assay, which initially received Emergency Use Authorization (EUA)…

On Oct. 5, 2020, Vaxxas announced a $22 million United States government award to support the deployment of…

On Oct. 5, 2021, the Nobel Prize in Chemistry 2020 was awarded to Benjamin List and David MacMillan…

On Oct. 5, 2020, Mateon Therapeutics announced that ArtiShieldTM had been approved for manufacture and marketing by the…

On Oct. 5, 2020, the Nobel Prize in Medicine 2020 was awarded to Harvey J. Alter, Michael Houghton…

On Oct. 5, 2020, the FDA awarded a research contract to the Stanford University School of Medicine to…

On Oct. 5, 2020, the CDC announced the existence of published reports that showed limited, uncommon circumstances where…

On Oct. 5, 2020, CLEAR, the secure identity company, and Quest Diagnostics announced a new collaboration that aimed…

On Oct. 5, 2020, CLEAR, the secure identity company, and LabCorp announced a partnership to enable users of…

On Oct. 5, 2020, Joe Verghese, M.B.B.S., M.S., an international leader in aging and cognition research at Albert…
On Oct. 5, 2020, the Fred Hutchinson Cancer Research Center announced the opening of the COVID-19 Clinical Research…

On Oct. 5, 2020, Bristol-Myers and MyoKardia announced a definitive merger agreement under which Bristol Myers Squibb will…

On Oct. 5, 2020, Corvus Pharmaceuticals announced that it has initiated a Phase 1 study to investigate a…

On Oct. 2, 2020, LabCorp announced that it had received an Emergency Use Authorization (EUA) from the FDA…

On Oct. 2, 2020, the Uconn Office of the Vice President for Research (OVPR) (OVPR) announced a new…
On Oct. 2, 2020, Yale scientists announced construction of the next-generation human brain PET scanner, the NeuroeXplorer (NX),…

On Oct. 1, 2020, the FDA approved Opdivo (nivolumab) in combination with Yervoy (ipilimumab) for the first-line treatment…
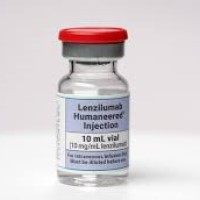
On Oct. 2, 2020, Humanigen announced that a patient case report on the use of lenzilumabル in critical…
On Oct. 2, 2020, Quidel announced that it had received Emergency Use Authorization from the U.S. Food and…

On Oct. 2, 2020, the WHO and the Kingdom of Saudi Arabia signed a contribution agreement in the…

On Oct. 2, 2020, AstraZeneca announced the phase I/II clinical trial for the COVID-19 vaccine AZD1222 had resumed…

On Oct. 2, 2020, the National Science Foundation announced investments in 24 new projects to drive future manufacturing….

On Oct. 1, 2020, Gilead Sciences announced that it will be responsible for distributing Veklury in the U.S….

On Oct. 1, 2020, widespread accurate testing for SARS-CoV-2, the virus that causes COVID-19, is one critical aspect…

On Oct. 1, 2020, Amyris announced that it has begun delivering samples to pharmaceutical companies of a sustainable…

On Oct. 1, 2020, Johnson & Johnson announced it had successfully completed its acquisition of Momenta Pharmaceuticals, a…
On Oct. 1, 2020, american hospitals began purchasing Veklury (remdesivir) directly from the drugメs distributor. Veklury is an…

On Sept. 30, 2020, Pfizer and BioNTech announced that preliminary, peer-reviewed data from their ongoing German Phase 1/2…

On Sept. 30, 2020, researchers from the Perelman School of Medicine at the University of Pennsylvania reported results…
On Sept. 30, 2020, BD (Becton, Dickinson) announced its rapid, point-of-care, SARS-CoV-2 antigen test for use on the…