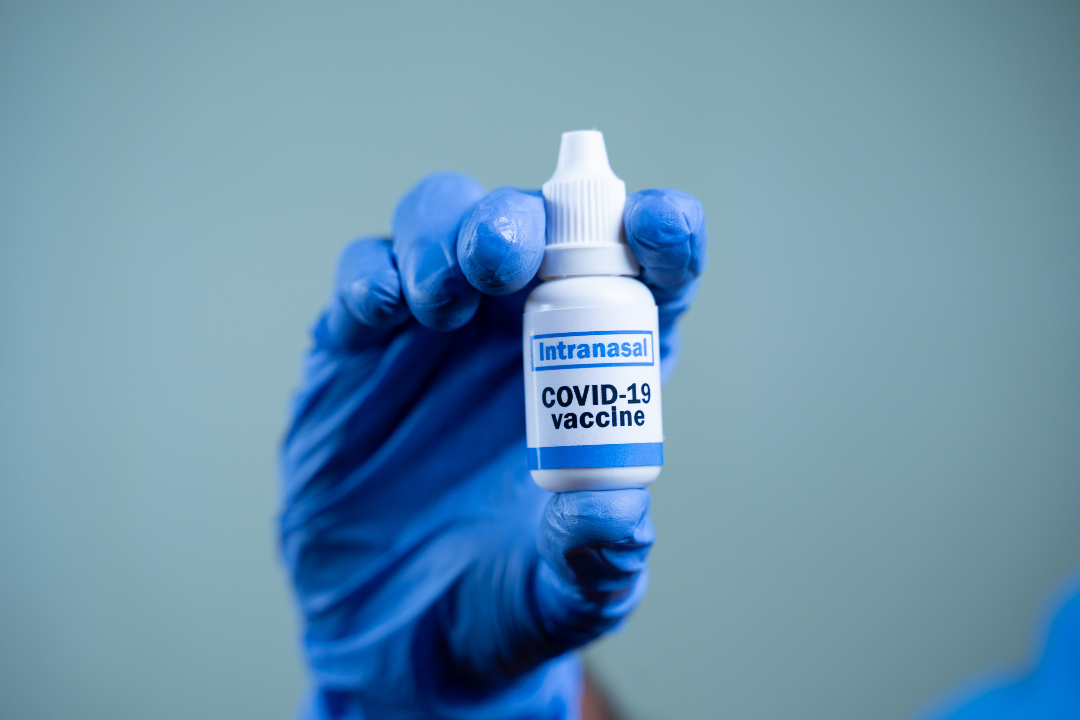
Altimmune Completed First Development Milestone Toward a Single-Dose Intranasal COVID-19 Vaccine
On Feb. 28, 2020, Altimmune announced the advancement of a novel single-dose, intranasal vaccine using Altimmune’s proprietary technology to protect against COVID-19, the disease caused by the SARS-CoV-2 virus. Altimmune had completed the design and synthesis of the vaccine and was advancing it toward animal testing and manufacturing.
The new intranasal vaccine is based on the same platform vaccine technology as NasoVAX, the company’s influenza vaccine candidate. Like NasoVAX, the COVID-19 vaccine is designed to provide systemic immunity following a single intranasal dose.
A recent Phase 2 clinical study with NasoVAX highlighted the ability of intranasal vaccine delivery to stimulate a durable and broad immune response against the influenza virus.
Altimmune believes the clinical profile of NasoVAX is particularly relevant to COVID-19 because intranasal delivery directs the immune response where it is needed most to protect against respiratory infection, including the likely site of initial viral attack. Clinical testing of the vaccine could begin as early as August.
Altimmune is a biopharmaceutical company focused on developing treatments for liver disease, immune modulating therapies and vaccines. Their diverse pipeline includes next generation peptide therapeutics for NASH (ALT-801) and chronic hepatitis B (HepTcell™), conjugated immunostimulants for the treatment of cancer (ALT-702) and intranasal vaccines (NasoVAX™ and NasoShield™).
Tags:
Source: Altimmune
Credit:
