University of British Columbia researchers unveiled first molecular images of B.1.1.7 COVID-19 mutation
On May 3, 2021, University of British Columbia (UBC) researchers were the first in the world to publish structural images of the N501Y mutation on the SARS-CoV-2 spike protein-a change believed to be partly responsible for the highly infectious nature and rapid spread of variant B.1.1.7. The pictures, taken at near-atomic resolution, provided critical insight as to why the B.1.1.7 variant ‘first detected in the U.K and accounted for a growing number of cases across Canad ‘ was more infectious.
UBC researchers are the first in the world to publish structural images of the N501Y mutation on the SARS-CoV-2 spike protein—a change believed to be partly responsible for the highly infectious nature and rapid spread of variant B.1.1.7. The pictures, taken at near-atomic resolution, provide critical insight as to why the B.1.1.7 variant—first detected in the U.K and now accounting for a growing number of cases across Canada—is more infectious. The images also add to the growing body of data indicating that existing vaccines are likely to remain effective in preventing mild and severe cases caused by B.1.1.7. Dr. Sriram Subramaniam, professor in UBC faculty of medicine’s department of biochemistry and molecular biology, discusses the implications of his team’s research, recently published in PLOS Biology.
Viruses are constantly mutating. The B.1.1.7 variant of concern, which was first reported to the World Health Organization in mid-December 2020, has an unusually large number of mutations. Of particular interest is a mutation known as N501Y located on the virus’s spike protein, which is what the virus uses to attach itself to human cells.
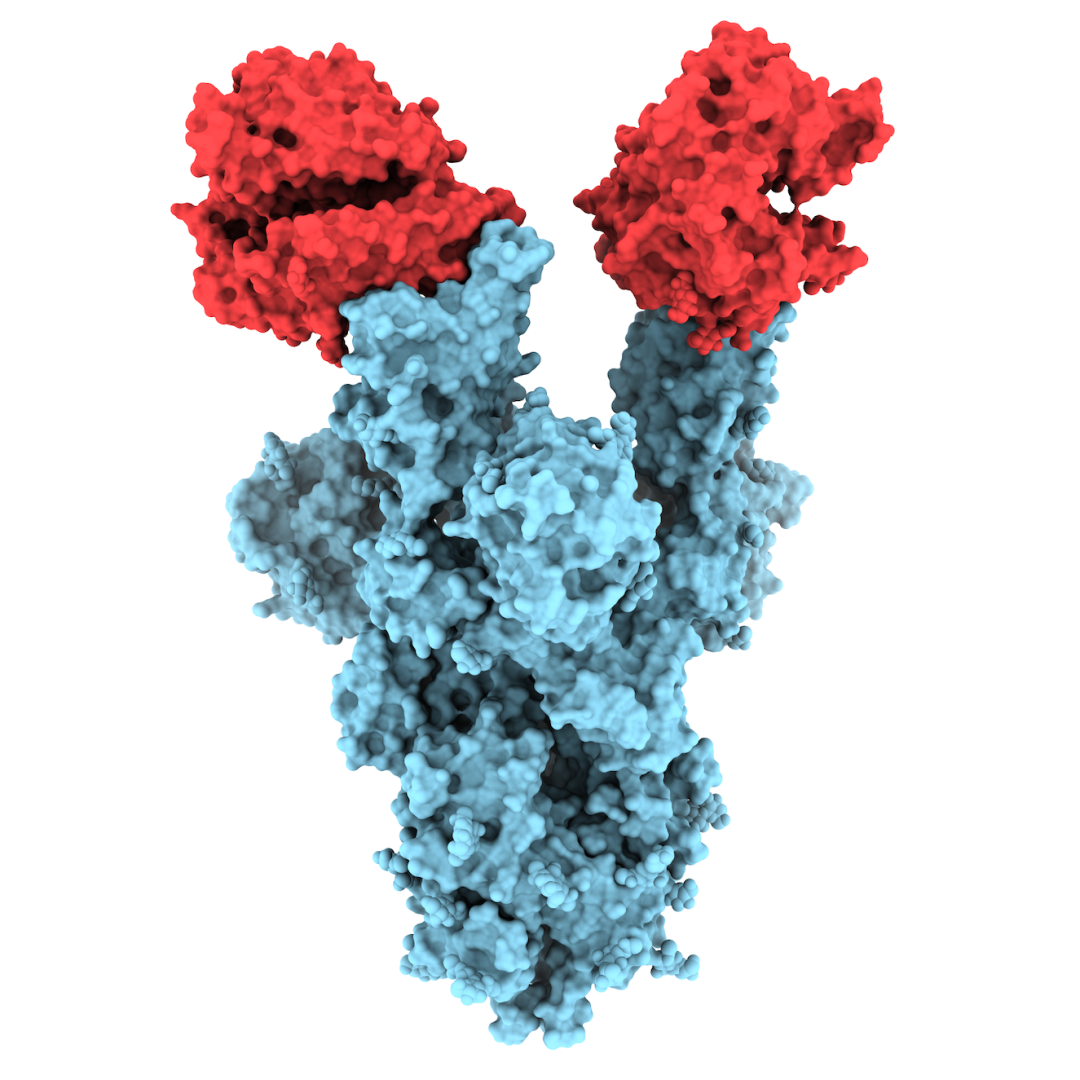
The images we captured provide the first structural glimpse of the N501Y mutant and show that the changes resulting from the mutation are localized. In fact, the N501Y mutation is the only mutation in the B.1.1.7 variant that is located on the portion of the spike protein that binds to the human ACE2 receptor, which is the enzyme on the surface of our cells that serves as the entry gate for SARS-CoV-2.
Our analysis revealed that even though the N501Y mutant can bind and enter our cells more readily, it can still be neutralized by antibodies that block the entry of the unmutated version of the virus into cells. This is an important observation and adds to the growing body of evidence that the majority of antibodies elicited in our immune system by existing vaccines are likely to remain effective in protecting us against the B1.1.7 variant.
Tags:
Source: University of British Columbia
Credit: Microscopy: N501Y spike protein mutant, shown (in blue) bound to two copies of the ACE2 receptor (in red), courtesy of the University of British Columbia.
