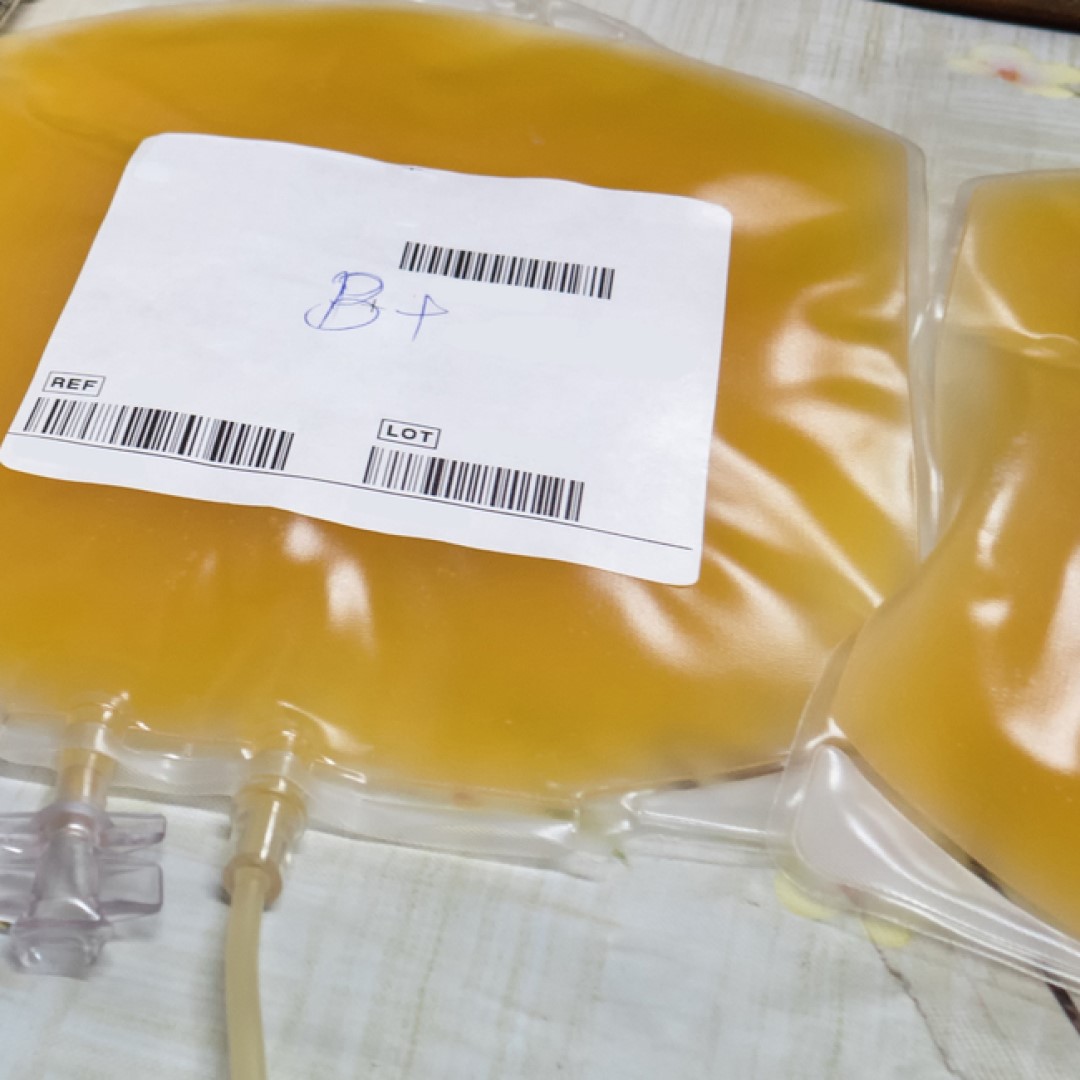
CIRM Board funded first clinical study for COVID-19
On Apr. 24, 2020, the governing Board of the California Institute for Regenerative Medicine (CIRM) awarded $749,999 to Dr. John Zaia at City of Hope. He will be conducting a clinical study to administer blood plasma from recovered COVID-19 patients to treat those with the virus. This marked CIRM’s first clinical study for COVID-19 after approving emergency funding a month earlier.
Plasma is a component of blood that carries proteins called antibodies that are usually involved in defending our bodies against viral infections. Blood plasma from patients that have recovered from COVID-19, referred to as convalescent plasma, contain antibodies against the virus that can be used as a potential treatment for COVID-19. Currently, there are challenges with this approach that include: properly identifying convalescent plasma donors i.e. recovered patients, determining eligibility of those with convalescent plasma that want to donate, collection of the plasma, treating patients, and determining if the plasma was effective.
Dr. Zaia and his team at City of Hope will create the COVID-19 Coordination Program, which addresses solutions for all of the challenges listed above. The program will partner with the medical teams at CIRM’s Alpha Stem Cell Clinic Network, as well as infectious disease, pulmonary and critical care teams from medical centers and community hospitals across the state. Potential donors will be identified and thoroughly screened for eligibility per the established National and State blood banking safety requirements. Finally, the convalescent plasma will be collected from eligible donors and administered by licensed physicians to COVID-19 patients, who will be evaluated for response to the treatment and potential recovery.
Tags:
Source: CIRM
Credit:
