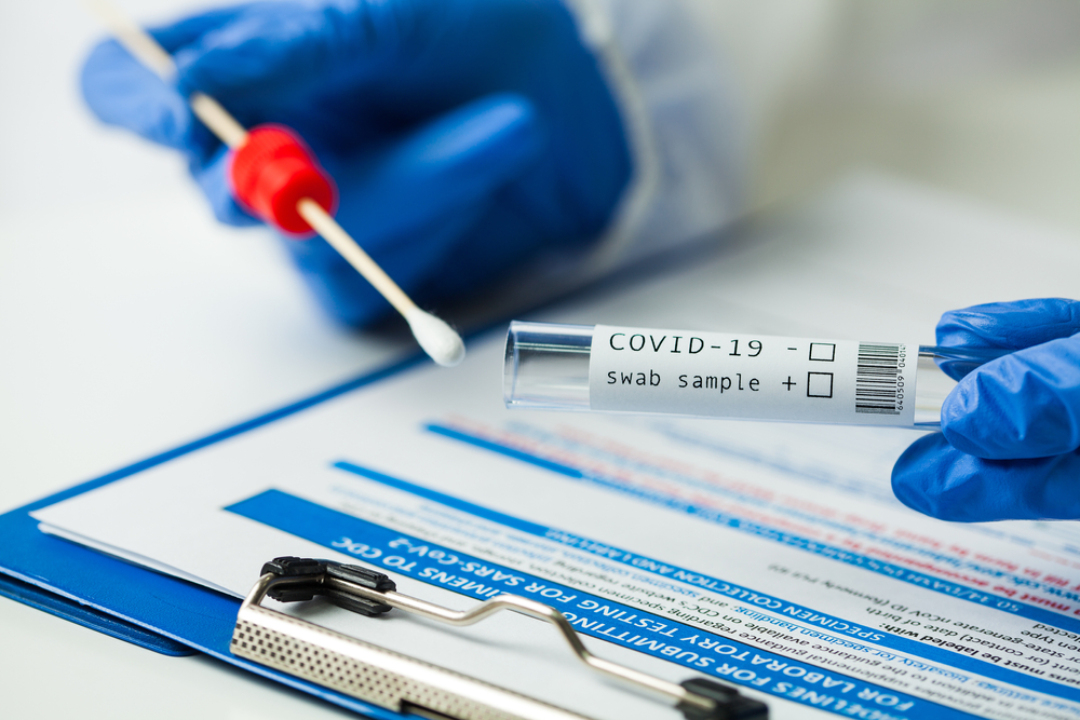
LabCorp launched test for Coronavirus (COVID-19)
On Mar. 5, 2020, LabCorp announced that it was making its LabCorp 2019 Novel Coronavirus (COVID-19), NAA test available immediately for ordering by physicians or other authorized healthcare providers anywhere in the U.S. The test detected the presence of the underlying virus that causes COVID-19 and was for use with patients who met current guidance for evaluation of infection with COVID-19.
The LabCorp 2019 Novel Coronavirus (COVID-19), NAA test was developed internally by LabCorp and is being made available pursuant to guidance issued by the U.S. Food and Drug Administration (FDA). LabCorp’s test has been validated for use with respiratory samples, including nasopharyngeal (NP) or oropharyngeal (OP) aspirates or washes, NP or OP swabs, and broncheoalveolar lavage (BAL). The test is a qualitative assay using PCR technology, which LabCorp played a central role in commercializing when PCR was introduced nearly 30 years ago.
LabCorp does not currently collect specimens for the test. Patients for whom testing has been ordered should not be sent to a LabCorp location to have a specimen collected. Instead, an appropriate specimen should be collected at the healthcare facility where the patient was seen and the test was ordered. The specimen should be sent to LabCorp using standard procedures. Test results will be available in 3-4 days.
Tags:
Source: LabCorp
Credit:
