CDC announced it had begun distribution of a new-generation smallpox vaccine
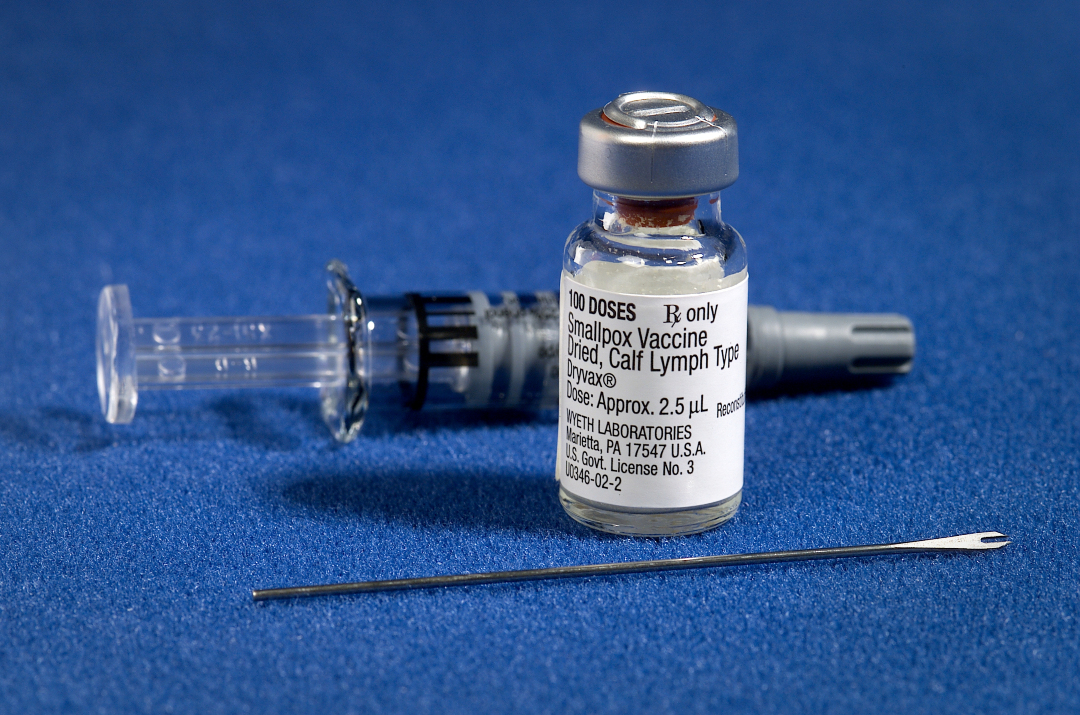
On Feb. 29, 2008, the U.S. Centers for Disease Control and Prevention (CDC) announced it had begun distribution of a new-generation smallpox vaccine, ACAM2000 (Acambis (acquired by Sanofi in July, 2008, Cambridge, Massachusetts), to civilian laboratory personnel, the military, and state public health preparedness programs.
Wyeth intends to withdraw the Dryvax license and asks that all remaining quantities of vaccine held by civilian and military users be quarantined by February 29, 2008, for the purpose of destruction.
This withdrawal is not necessitated by any safety, purity, or quality concerns with the product but rather is consistent with a contract agreement between CDC and Wyeth. All lots of Dryvax vaccine will expire on February 29, 2008, and should not be used after that date.
The CDC will continue to provide ACAM2000 smallpox vaccine to protect responders as part of state public health preparedness programs and civilian laboratory personnel who risk exposure to orthopoxviruses. Unlike Dryvax, ACAM2000 expires 18 months after release from the CDC Strategic National Stockpile.
Tags:
Source: U.S. Centers for Disease Control and Prevention
Credit:
