Pfizer and Valneva Announce Lyme Disease Vaccine Candidate Demonstrates Strong Efficacy in Phase 3 VALOR Trial
On Mar. 24, 2026, Pfizer and Valneva announced topline results from the Phase 3 VALOR “Vaccine Against Lyme for Outdoor Recreationists” clinical trial of its investigational 6-valent OspA-based Lyme disease vaccine candidate PF-07307405 (LB6V, formerly known as VLA15) demonstrating:
In the pre-specified analyses:
- Efficacy of 73.2% from 28 days post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 15.8, 93.5)
- Efficacy of 74.8% from 1-day post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 21.7, 93.9)
Fewer than anticipated Lyme disease cases were accrued over the study period, and the pre-determined statistical criterion (95% confidence interval lower bound >20) was not met in the first pre-specified analysis (primary endpoint). Given the clinically meaningful efficacy and the fact that the 95% confidence interval lower bound was above 20 in the second pre-specified analysis, Pfizer is confident in the vaccine’s potential and is planning submissions to regulatory authorities.
Developed in collaboration between Pfizer and Valneva, the investigational 6-valent OspA-based Lyme disease vaccine is being evaluated for its efficacy, safety, tolerability, immunogenicity, and manufacturing lot consistency. The companies entered into a collaboration and license agreement in April 2020 for the co-development of PF-07307405 and for Pfizer to exclusively manufacture and commercialize PF-07307405, assuming regulatory success.
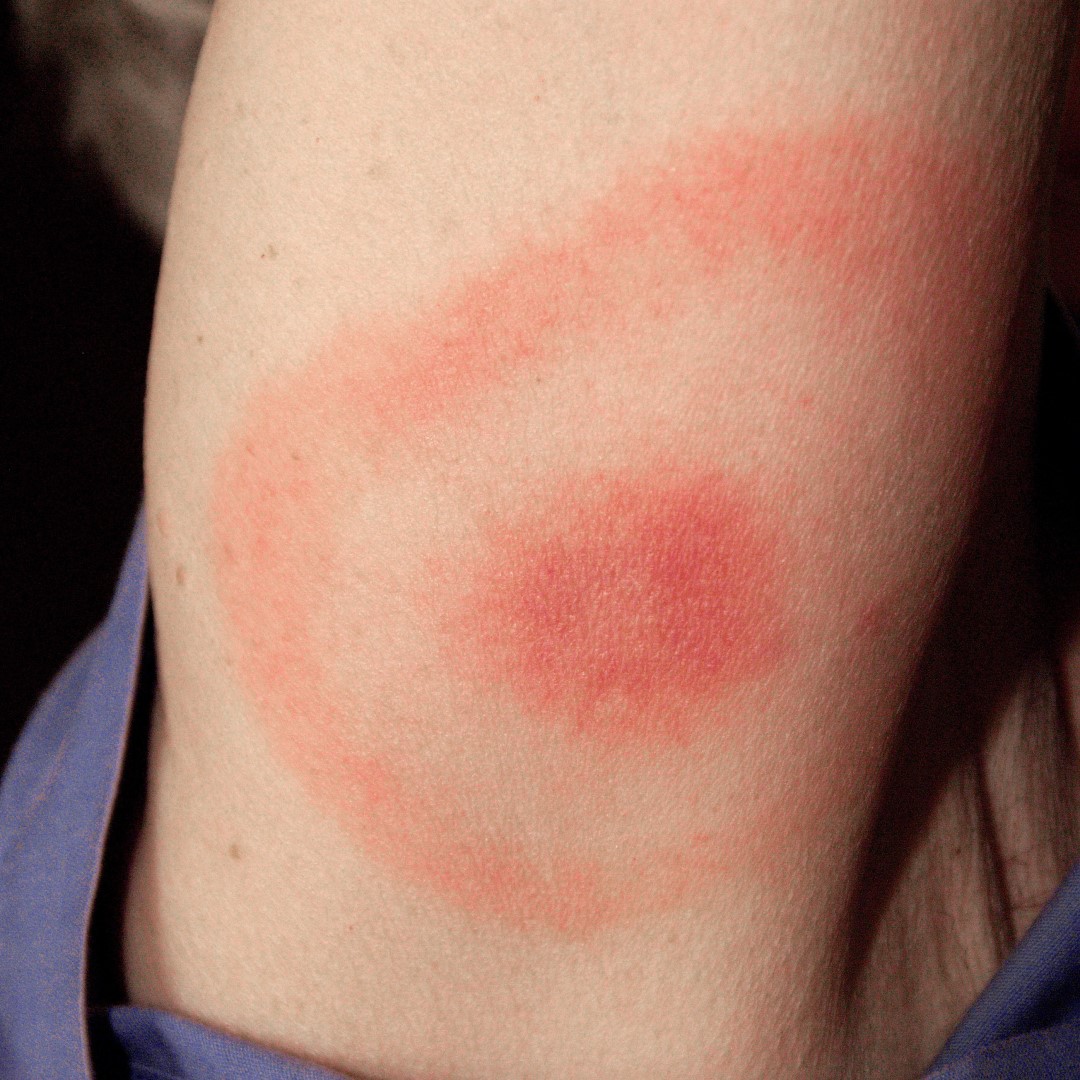
Lyme disease is a systemic infection caused by Borrelia burgdorferi sensu lato bacteria that are transmitted to humans by the bite of infected Ixodes ticks.It is considered the most common vector-borne illness in the Northern Hemisphere. The Centers for Disease Control and Prevention (CDC) has estimated that approximately 476,000 people in the U.S. are diagnosed and treated each year and 132,000 cases are reported annually in Europe from countries with surveillance systems.
Early symptoms of Lyme disease (such as a gradually expanding erythematous rash called erythema migrans or other nonspecific symptoms like fatigue, fever, headache, mild stiff neck, muscle and joint pains) are often overlooked or misinterpreted. Left untreated, the disease can disseminate and cause more serious chronic complications affecting the skin, joints (arthritis), the heart (carditis) or the nervous system. The medical need for vaccination against Lyme disease is steadily increasing as the geographic footprint of the disease widens.
Tags:
Source: Pfizer
Credit:
