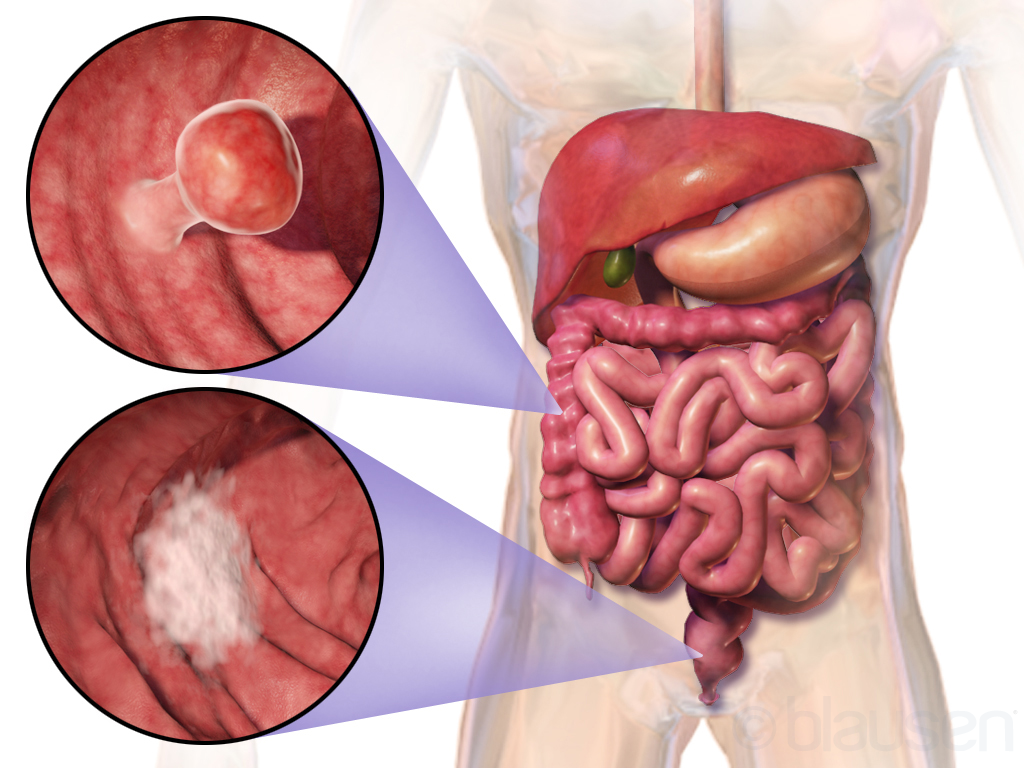
The FDA approved another topoisomerase 1 inhibitor for the treatment of metastatic colorectal cancer
In June 1996, the Food and Drug Administration (FDA) first approved irinotecan for the treatment of patients with metastatic carcinoma of the colon or rectum whose disease had recurred or progressed following 5-fluorouracil (5-FU)-based therapy (second-line therapy).
Colorectal cancer is the third most common cause of cancer mortality in the United States. It constitutes about 10% of worldwide cancer deaths.
Irinotecan is combined with 5-fluorouracil (5-FU) and leucovorin for maximum efficacy against colorectal cancer. It is considered more efficacious in combination with 5-FU/leucovorin than the separate individual use of the agents. This combination is also known as the FOLFIRI regimen. With therapy regimens like FOLFIRI, the median survival rate of a patient with metastatic colorectal cancer has improved from 8 months to 24 months.
Tags:
Source: U.S. National Library of Medicine
Credit:
