The NIH awarded up to $130 million over seven years for UGA to develop universal flu vaccine
On Sept. 30, 2019, the University of Georgia (UGA) announced it had signed a contract with the National…
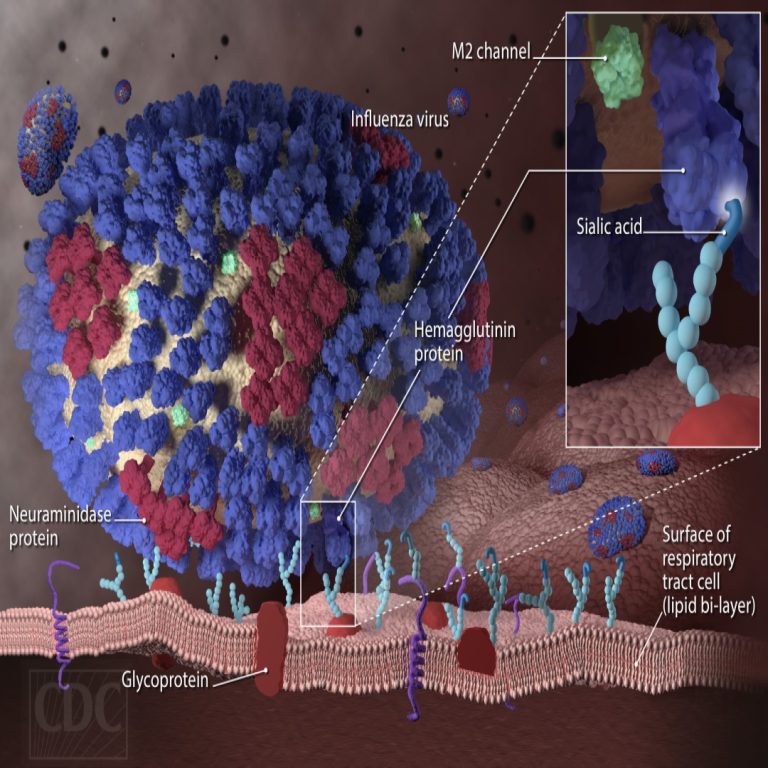
On Sept. 30, 2019, the University of Georgia (UGA) announced it had signed a contract with the National…
In Jun. 2019, Defense Advanced Research Projects Agency (DARPA) announced it had funded a $21.9 gene modulation research…

On Jun. 5, 2019, Vaccitech announced that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100)…
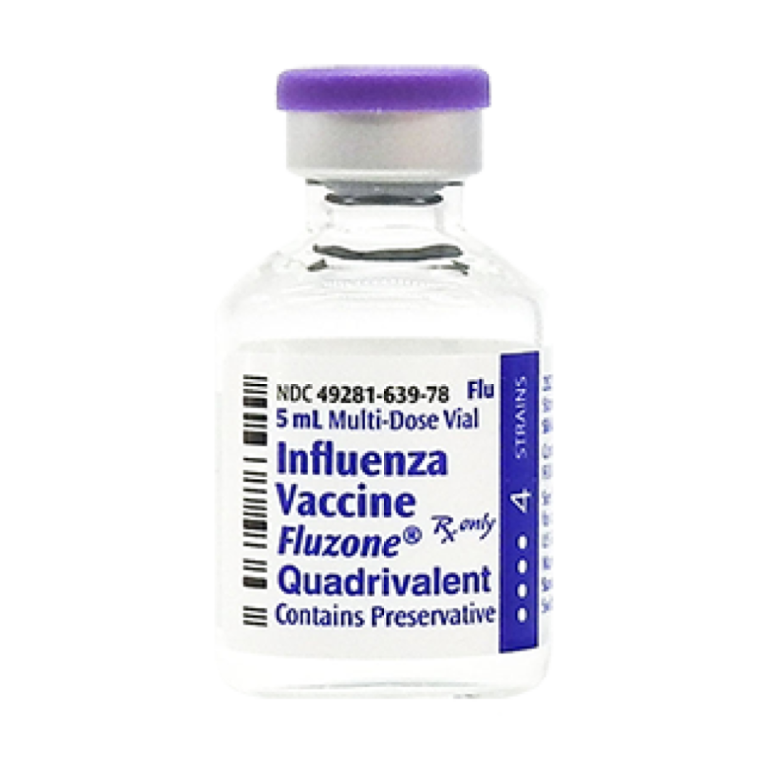
On Jan. 23, 2019, U.S. Food and Drug Administration (FDA) announced it had approved approved the use of…
On Aug. 24, 2018, the Centers for Disease Control and Prevention (CDC) published Immunization Practices Advisory Committee’s (ACIP)…
On Jun. 8, 2018, the U.S. Centers for Disease Control and Prevention (CDC) published Immunization Practices Advisory Committee…

On Apr. 23, 2018, Medigen Vaccine Biologics and GC Pharma entered an exclusive distribution agreement for GCC’s quadrivalent…

On Apr. 1, 2018, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices and the…
On Jan. 11, 2018, the U.S. Food and Drug Administration (FDA) approved an expanded indication for the four-strain…

On Aug. 25, 2017, the U.S. Centers for Disease Control and Prevention (CDC) published Immunization Practices Advisory Committee…
On Feb. 28, 2014, the U.S. Centers for Disease Control and Prevention (CDC) published Immunization Practices Advisory Committee…

On Dec. 14, 2012, the U.S. Food and Drug Administration (FDA) announced it had approved GlaxoSmithKline’s quadrivalent formulation…

On Nov. 20, 2012, the FDA approved a seasonal flu vaccine produced by Novartis using animal cell culture…
On Nov. 20, 2012, the U.S. Food and Drug Administration (FDA) announced it had approved first seasonal influenza…

On Aug. 10, 2010, WHO Director-General Dr Margaret Chan announced that the H1N1 influenza virus had moved into…

On Feb. 24, 2010, the Immunization Practices Advisory Committee (ACIP) recommended universal Influenza vaccination for those 6 months…
On Dec. 23, 2009, the FDA approved high-dose inactivated influenza vaccine (Fluzone High-Dose) for people ages 65 years…

On Oct. 24, 2009, President Barack Obama declared the swine flu (N1n1) outbreak a national emergency. The declaration…

On Jul. 31, 2009, the Immunization Practices Advisory Committee (ACIP) updates the 2008 recommendations regarding the use of…
On Jun. 11, 2009, the Dr. Margaret Chan, Director-General World Health Organization (WHO), declared the world was at…

On May 22, 2009, the U.S. Dept. of Health and Human Services (HHS) directed $1 billion toward development…

On Apr. 15, 2009, the U.S. Centers for Disease Control and Prevention (CDC) identified the novel H1N1 influenza…

In Apr. 2009, physicians used point of care rapid immunoassay tests to provide influenza results within 15 minutes…
In 2009, an increased risk of narcolepsy (a chronic sleep disorder) was found following vaccination with Pandemrix, a…

On Jun. 23, 2008, Sanofi Pasteur announced the U.S. Food and Drug Administration (FDA) had licensed Pentacel, Diphtheria…
On Feb. 27, 2008, the Immunization Practices Advisory Committee (Immunization Practices Advisory Committee (ACIP) voted to expand influenza…

In 2008, the U.S. Centers for Disease Control and Prevention (CDC) received U.S. Food and Drug Administration (FDA)…

In 2008, the Influenza Reagent Resource (IRR) was established by the Centers for Disease Control and Prevention (CDC)…

On Sept. 20, 2007, the U.S. Food and Drug Administration (FDA) approved use of FluMist nasal-spray influenza vaccine…

On Jun. 7, 2007, years before the COVID-19 pandemic swept across the globe, a group of about 100…