Meridian Bioscience supports companies in fighting Coronavirus withg zero cost proprietary technology for evaluation
On Mar. 16, 2020, Meridian Bioscience announced its increased commitment to the global fight against COVID-19 by making…

On Mar. 16, 2020, Meridian Bioscience announced its increased commitment to the global fight against COVID-19 by making…

On Mar. 15, 2020, Iceland health authorities and deCode Genetics announced they began comprehensive screening for the virus…
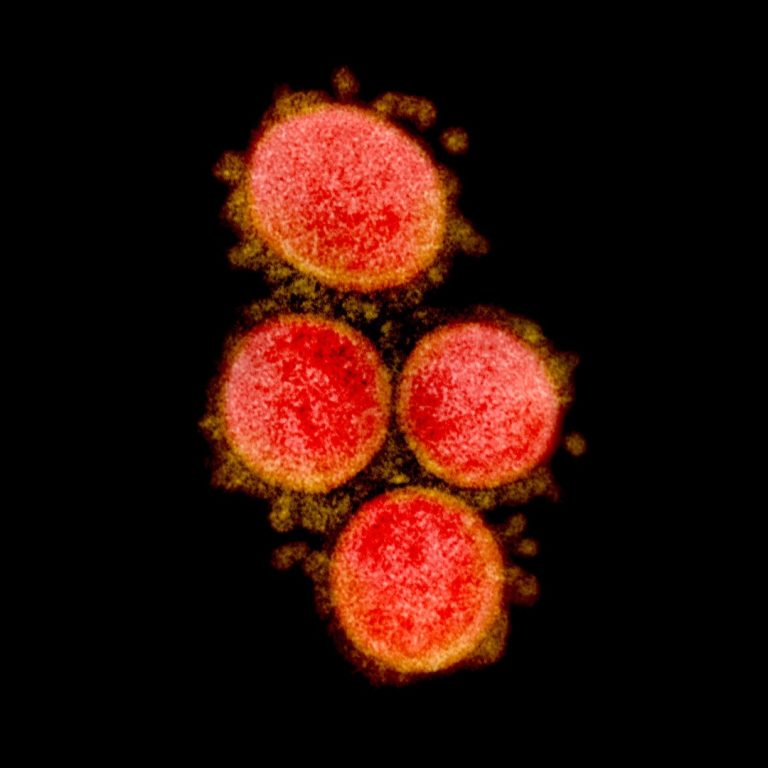
On Mar. 15, 2020, the National Institutes of Health (NIH) informed its staff that it had its first…
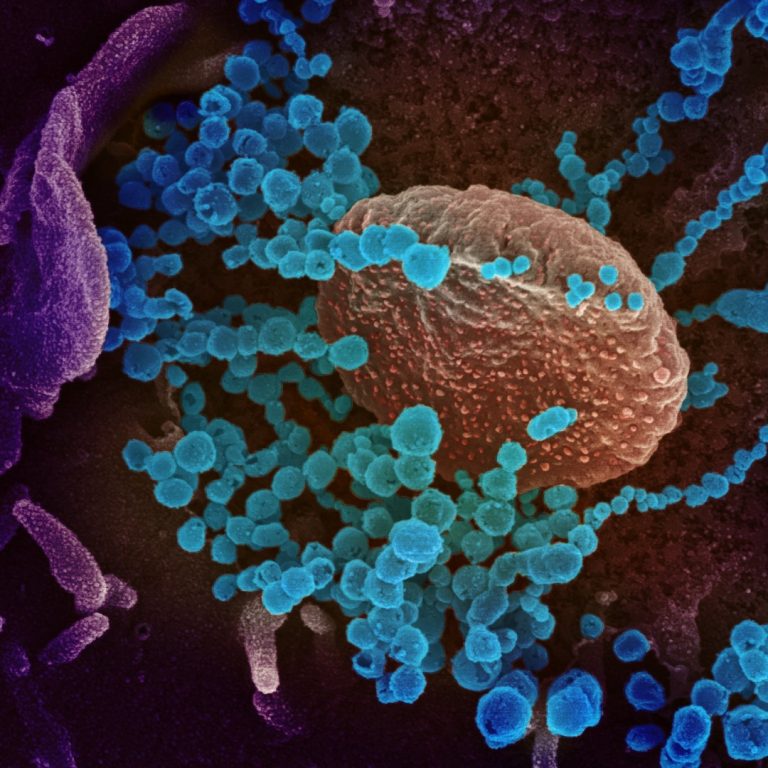
On Mar. 13, 2020, Enanta Pharmaceuticals announced it had initiated a program to discover direct-acting antiviral drug candidates…

On Mar. 13, 2020, researchers at the National Eye Institute (NEI) reported finding a protein that normally deposits…

On Mar. 13, 2020, declared a COVID-related emergency under Section 501(b) of the Stafford Act [18]. Under the…

On Mar. 12, 2020, Pluristem Therapeutics announced it a collaborative agreement with the BIH Center for Regenerative Therapy…
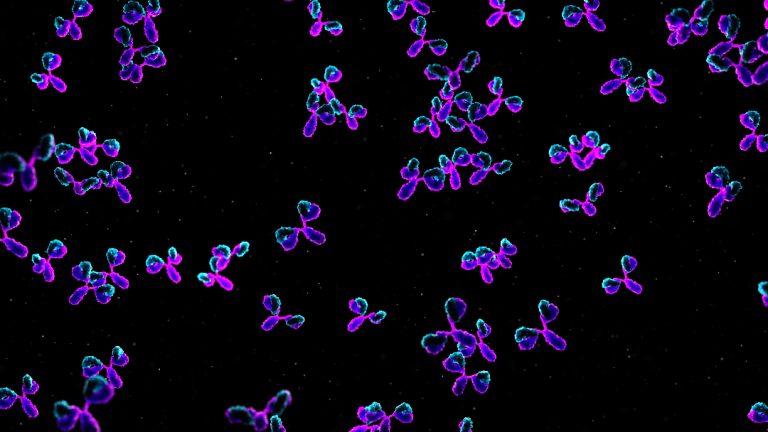
On Mar. 12, 2020, Vir Biotech announced it had signed a letter of intent with Biogen for the…

On Mar. 12, 2020, Emory University announced it was participating in a National Institutes of Health (NIH) sponsored…

On Mar. 12, 2020, AbCellera and Eli Lilly announced an agreement to co-develop antibody products for the treatment…

On Mar. 12, 2020, Innovation Pharma announced research procedures that one of the 12 Regional Biocontainment Labs (RBLs)…

On Mar. 12, 2020, Predictive Oncology announced it will launch a new AI platform for vaccine and drug…

On Mar. 11, 2020, the antiretroviral drugs dolutegravir and emtricitabine/tenofovir alafenamide fumarate (DTG+FTC/TAF) may comprise the safest and…

On Mar. 11, 2020, the Howard Hughes Medical Institute (HHMI) announced a new investment of over $120 million…

On Mar. 11, 2020, the La Jolla Institute for Immunology (LJI) and the J. Craig Venter Institute announced…

On Mar. 11, 2020, Emergent BioSolutions announced it has initiated development of two product candidates for treatment and…

On Mar. 10, 2020, the Bill & Melinda Gates Foundation, Wellcome, and Mastercard committed up to $125 million…

On Mar. 10, 2020, Innovation Pharma announced that management was advised that testing of Brilacidin, the Company’s flagship…

On Mar. 9, 2020, Winship Cancer Institute’s multiple myeloma team was awarded a $5 million grant from The…
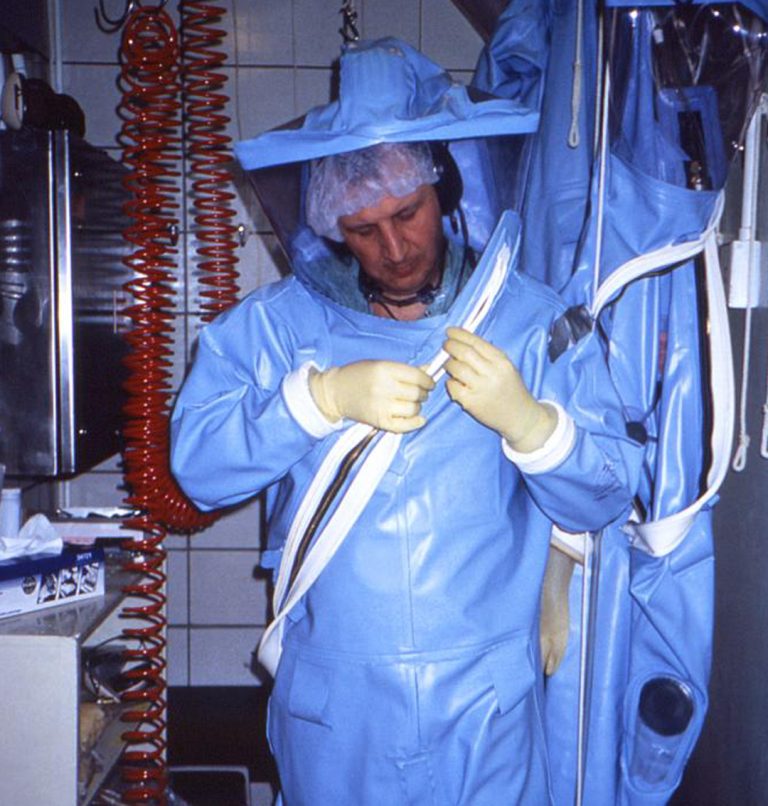
On Mar. 9, 2020, Innovation Pharmaceuticals announced it was notified by one of the 12 Regional Biocontainment Labs…

On Mar. 9, 2020, AIM ImmunoTech announced that the National Institute of Infectious Diseases (NIID) in Japan will…

On Mar. 9, 2020, The U.S. Food and Drug Administration (FDA) approved Ofev (nintedanib) oral capsules to treat…
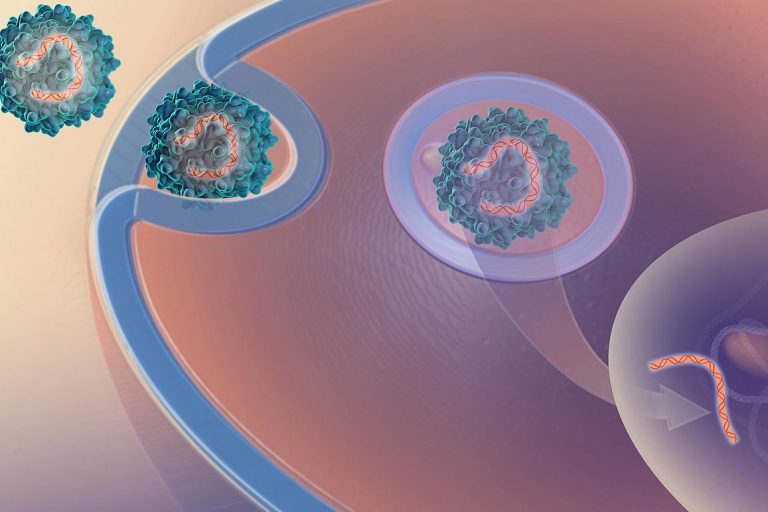
On Mar. 9, 2020, researchers from the National Institute of Allergy and Infectious Diseases (NIAID), part of the…
On Mar. 9, 2020, MediciNova announced that it planned to initiate development of MN-166 (ibudilast) for severe pneumonia…

On Mar. 8, 2020, an international team of investigators reported finding a protein produced by the human immune…

On Mar. 6, 2020, the U.S. Food and Drug Administration (FDA) approved Isturisa (osilodrostat) oral tablets for adults…

On Mar. 6, 2020, to further contribute to global efforts to address the COVID-19 outbreak, Canada’s Minister of…

On Mar. 6, 2020, University of British Columbia (UBC) researchers Yue Qian, Dr. Richard Lester, Dr. Srinivas Murthy,…

On Mar. 6, 2020, Innovation Pharmaceuticals continues to receive inquiries regarding the potential of Brilacidin, the Company’s novel…
On Mar. 6, 2020, Mount Sinai Health System and Harbour BioMed (HBM) announced a multi-year, multifaceted collaboration to…