The USDA confirmed COVID-19 in Hyenas at a Colorado Zoo
On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, Pfizer announced it was investigational novel COVID-19 oral antiviral candidate, PAXLOVID, significantly reduced hospitalization…

On Nov. 5, 2021, Chugai Pharmaceutical, announced that it had obtained approval from the Ministry of Health, Labour…

On Oct. 29, 2021, Oregon Health & Science University’s (OHSU) board of directors approved a project to expand…

On Oct. 28, 2021, Pfizer and BioNTech announced that the U.S. government had purchased 50 million more doses…

On Oct. 25, 2021, Moderna announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human…

On Oct. 21, 2021, Pfizer and BioNTech announced topline results from a Phase 3 randomized, controlled trial evaluating…

On Oct. 20, 2021, Quest Diagnostics announced it had formed an agreement with the Texas Department of State…
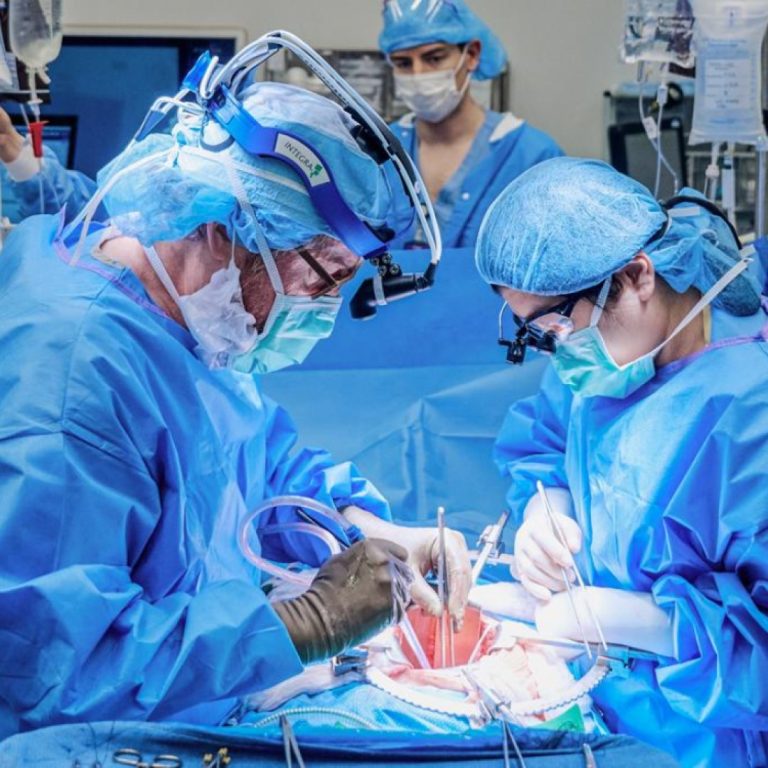
On Oct. 19, 2021, the first investigational transplant of a genetically engineered, nonhuman kidney to a human body…

On Oct. 18, 2021, Pfizer and BioNTech announced that the European Medicines Agency’s Committee for Human Medicinal Products…

On Oct. 18, 2021, Gilead Sciences announced that the Food and Drug Administration had approved a new low-dose…

On Oct. 18, 2021, Athenex and the Center for Cell and Gene Therapy at Baylor College of Medicine,…

On Oct. 15, 2021, Roche announced that the U.S. Food and Drug Administration (FDA) had approved Tecentriq (atezolizumab)…

On Oct. 15, 2021, Pfizer and BioNTech announced they had submitted data supporting the vaccination of children 5…

On Oct. 13, 2021, the World Health Organization (WHO) honoured the late Henrietta Lacks with a WHO Director-General’s…

On Oct. 12, 2021, National Resilience announced a strategic collaboration with Children’s Hospital of Philadelphia (CHOP) to implement…

On Oct. 11, 2021, scientists at Washington University School of Medicine in St. Louis announced they had developed…
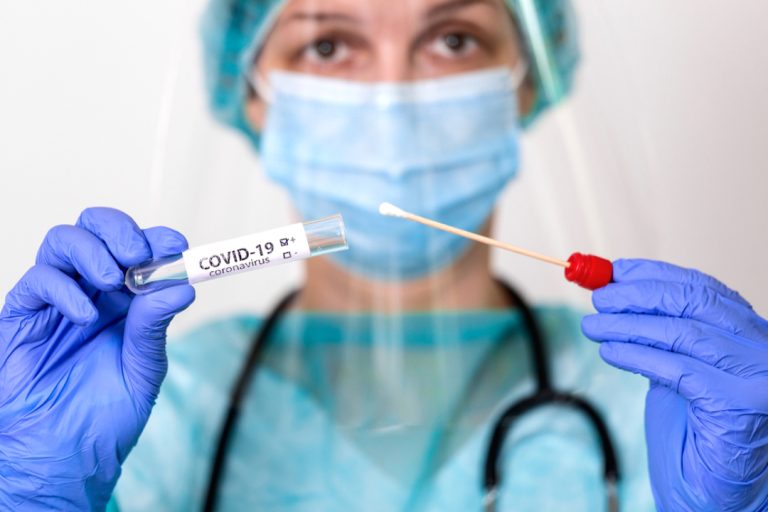
On Oct. 7, 2021, PerkinElmer announced that the U.S. Food and Drug Administration (FDA) had issued Emergency Use…

On Oct. 4, 2021, the Fred & Pamela Buffett Cancer Center announced it had successfully renewed its National…

On Oct. 4, 2021, Pfizer and BioNTech announced that the Committee for Medicinal Products for Human Use (CHMP)…

On Oct. 1, 2021, Merck and Ridgeback Biotherapeutics announced that molnupiravir (MK-4482, EIDD-2801), an investigational oral antiviral medicine,…
This cartoon illustrates the various issues facing individuals across the U.S. who are vaccinated against COVID-19 verus those…

On Sept. 29, 2021, Pfizer and BioNTech announced they had submitted data to the U.S. Food and Drug…
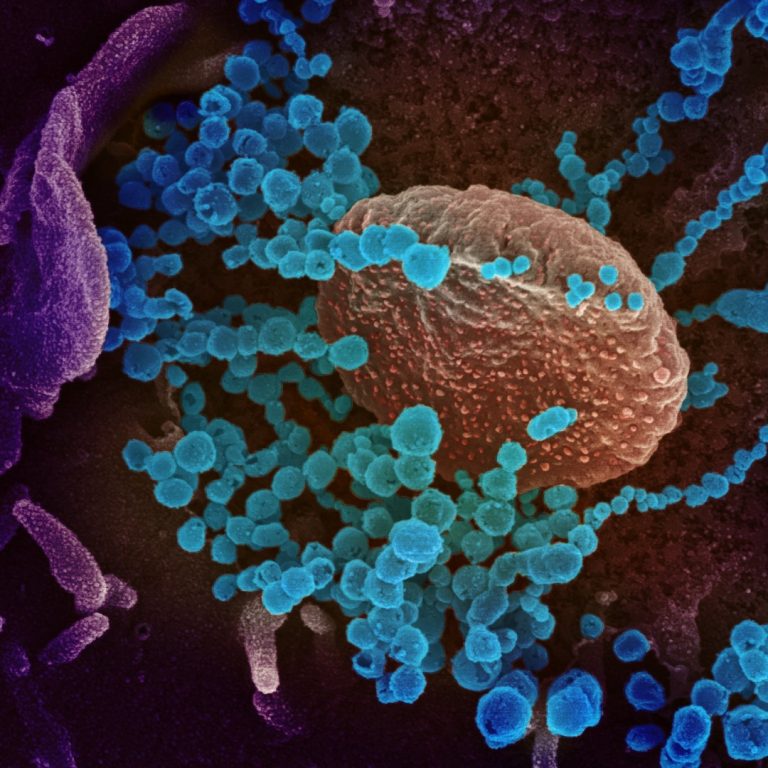
On Sept. 29, 2021, Regeneron announced that the New England Journal of Medicine (NEJM) published positive detailed results…

On Sept. 29, 2021, Roche confirmed positive data from the phase II/III 2066 study, investigating Ronapreveル (casirivimab and…

On Sept. 29, 2021, the University of Pennsylvania announced that the Breakthrough Prize in Life Sciences was awarded…

On Sept. 28, 2021, Soligenix announced publication of pre-clinical immunogenicity studies for CiVax (heat stable COVID-19 vaccine program)…

On Sept. 23, 2021, Tonix Pharmaceuticals announced it had expanded its research collaboration with Columbia University. The research…

On Sept. 22, 2021, Gilead Sciences announced positive results from a Phase 3 randomized, double-blind, placebo-controlled trial to…

On Sept. 22, 2021, Pfizer and BioNTech announced plans to expand their agreement with the U.S. government by…