Scripps Scientists build an “evolution engine” to rapidly reprogram proteins
On Aug. 7, 2025, research scientists at Scripps Research announced a new platform that enables fast, scalable protein…
On Aug. 7, 2025, research scientists at Scripps Research announced a new platform that enables fast, scalable protein…

On Jul. 30, 2025, a study led by Northwestern Medicine shows that most U.S. adults have a “heart…

On Jul. 25, 2025, Simtra BioPharma announced the purchase from Cook Group of a 65-acre property (301 N….

On Jun. 30, 2025, as in something out of science fiction, a team of researchers reached into the…

On Jun. 20, 2025, the US Food and Drug Administration (FDA) announced it has approved Dupixent (dupilumab) for…

On Jun. 18, 2025, Gilead Sciences announced that the U.S. Food and Drug Administration (FDA) has approved Yeztugo®…

On Jun. 18, 2025, SandboxAQ, an artificial intelligence startup spun out of Alphabet’s Google and backed by Nvidia…
On Jun. 16, 2025, the National Institute for Health and Care Excellence (NICE) announced that the recommendation in…
On Jun. 13, 2025, people in England will become the first in the world to receive belantamab mafodotin…

On Jun. 4, 2025, the Conference of State Parties (CoSP) of the African Medicines Agency (AMA) announced the…
On May 22, 2025, Sanofi announces the formal opening of the company’s new flagship U.S. offices, unveiling a…

On May 20, 2025, the World Health Organization (WHO) announced that world leaders pledged at least an additional…

On May 19, 2025, Prime Medicine announced positive initial data from the first patient dosed in its ongoing…

On May 9, 2025, a McGill University led team of researchers announced a study that shows an estimated…

On May 7, 2025, researchers at the University College London (UCL) and King’s College London announced that an…

On May 5, 2025, Bristol Myers Squibb said it will invest $40 billion in the U.S. over the…

On Apr. 22, 2025, Regeneron Pharmaceuticals announced a significant expansion of its manufacturing capacity through a new agreement…

On Apr. 10, 2025, Novartis, a leading global innovative medicines company, announced a planned $23 billion investment over…

On Apr. 1, 2025, layoff notices began arriving for thousands of employees of the sprawling U.S. Department of…

On Mar. 26, 2025, the Biotechnology Innovation Organization (BIO) released results from a membership survey that underscores the…

On Mar. 7, 2025, a team of researchers from Aalto University and the University of Bayreuth announced that…

On Mar. 3, 2025, LifeScienceHistory.com announced the release of “Earth is a Rock, Let the DNA Mold Me”…

On Mar. 3, 2025, Genentech announced that the U.S. Food and Drug Administration (FDA) has approved TNKase® (tenecteplase),…
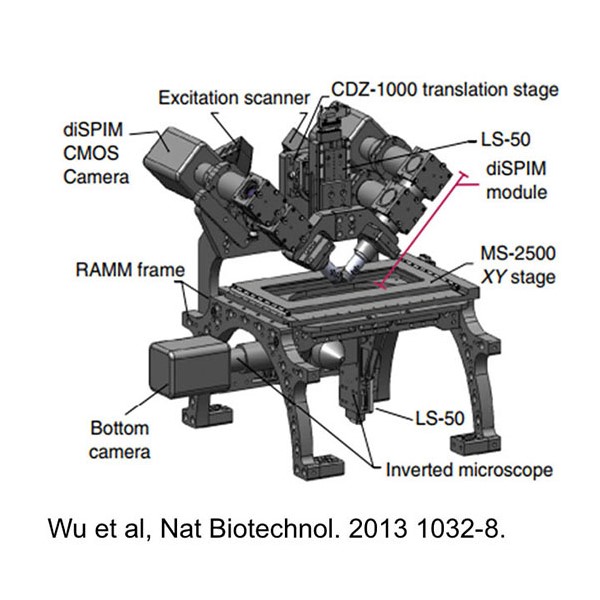
On Feb. 21, 2025, a team of researchers from the Marine Biological Laboratory (MBL) announced a hybrid custom-designed…
Jan. 24, 2025, the U.S. Food and Drug Administration (FDA) pulled draft guidance from its website requiring companies…
On Jan. 22, 2025, scientists at deCODE genetics/Amgen announced they have constructed a complete map of how human…
On Jan. 21, 2025, Rice University announced that four research groups are part of an inaugural cohort of…

On Jan. 14, 2025, the U.S. Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) announced…

On Jan. 10, 2025, the Institute for Health Metrics and Evaluation (IHME) reported that when it comes to…

On Dec. 11, 2024, Massachusetts General Hospital (MGH), founding member of the Mass General Brigham healthcare system, announced…