International community rallied to support open research and science to fight COVID-19
On May 29, 2020, the World Health Organization (WHO) announced that thirty countries and multiple international partners and…
On May 29, 2020, the World Health Organization (WHO) announced that thirty countries and multiple international partners and…

On May 29, 2020, the California Institute for Regenerative Medicine (CIRM) approved investments in three early-stage research programs…

On May 29, 2020, Moderna announced the first participants in each age cohort had been dosed in the…
On May 29, 2020, Johnson & Johnson announced that its Janssen Pharmaceutical subsidiary received a positive opinion from…
On May 27, 2020, the National Institutes of Health (NIH) announced it had found that molecules released into…

On May 27, 2020, Gilead Sciences and Arcus Biosciences announced that the companies had entered into a 10-year…

On May 27, 2020, an analyses of the largest publicly available catalog of human genomic data revealed new…

On May 27, 2020, Novavax announced the acquisition of Praha Vaccines, part of the Cyrus Poonawalla Group, in…

On May 26, 2020, Sanofi announced that it had agreed to sell 11.8 million shares of Regeneron common…
On May 26, 2020, Novavax announced enrollment of the first participants in a Phase 1/2 clinical trial of…

On May 26, 2020, Todos Medical announced it had entered into a cross-distribution agreement with L1 Systems, a…

On May 26, 2020, the Rice University COVID-19 Research Fund Oversight and Review Committee announced it had awarded…

On May 22, 2020, COVID 19 disrupted life-saving immunization services around the world, putting millions of children –…

On May 22, 2020, the California Institute for Regenerative Medicine (CIRM) board expanded efforts related to the $5…

On May 21, 2020, Abbott announced an interim analysis of an ongoing multi-site clinical study in urgent care…

On May 21, 2020, AstraZeneca announced it was advancing its response to address the unprecedented challenges of COVID-19,…

On May 21, 2020, BioAegis Therapeutics announced that the National Institute of Health Clinical Center began measuring patient…

On May 21, 2020, Roche announced that it had acquired Seattle-based Stratos Genomics, an early-stage sequencing technology company…

On May 20, 2020, Mammoth Biosciences announced a collaboration with GSK Consumer Healthcare to develop an accurate, easy-to-use,…

On May 20, 2020, BioSig Technologies and its subsidiary, ViralClear Pharmaceuticals, announced the closing of a $10.8 million…

On May 20, 2020, CHF Solutions announced a publication highlighting the use of ultrafiltration for fluid management of…
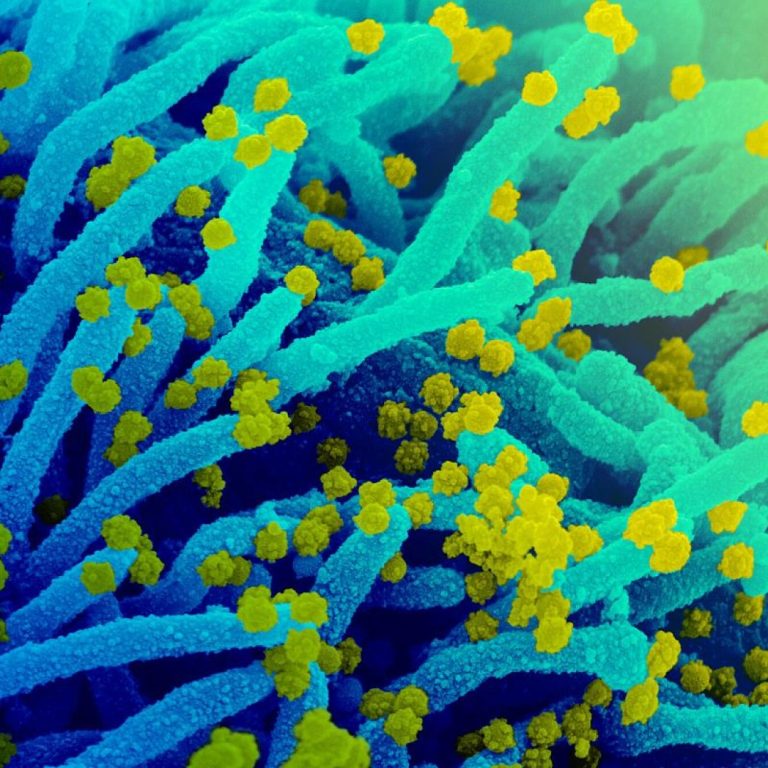
On May 20, 2020, Airway Therapeutics and Celonic Group announced a collaboration to produce the novel human recombinant…

On May 20, 2020, INOVIO announced the publication of the preclinical study data for IN0-4800, its COVID-19 DNA…

On May 19, 2020, the University of Oxford announced the release of the T-SPOT Discovery SARS-CoV-2 test kit….

On May 19, 2020, a University of Southern California (USC) study reported results from the first phase of…
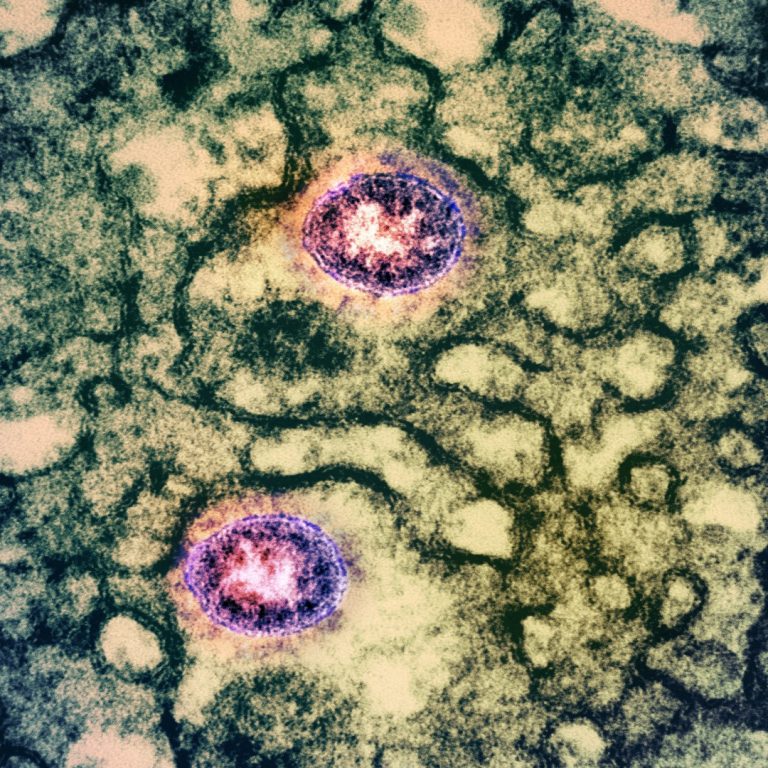
On May 19, 2020, in a study published in Cell Reports, researchers at the University of Hong Kong…

On May 19, 2020, Phlow, a U.S.-based, public benefit drug manufacturing corporation, received federal government funding of $354…
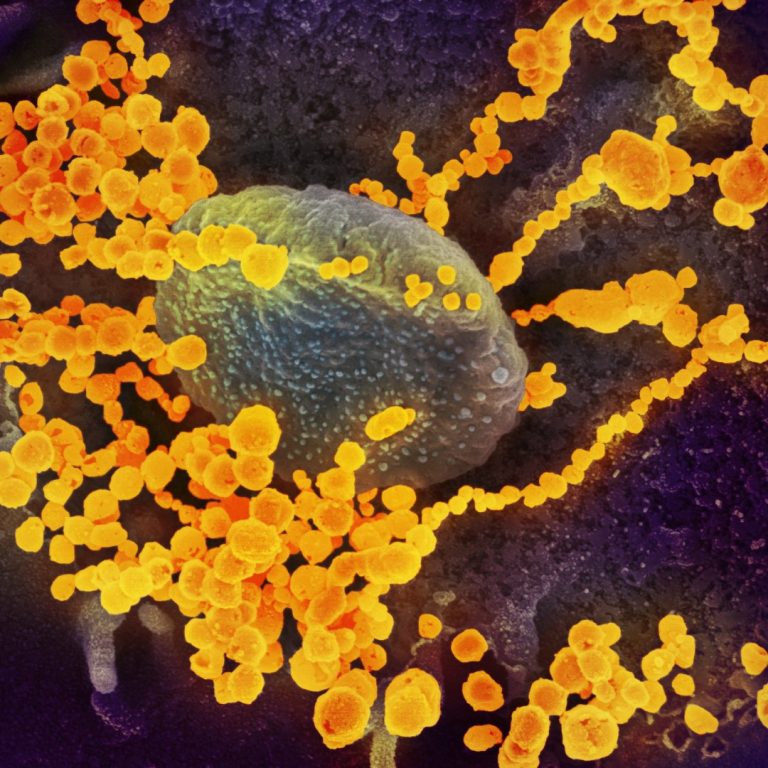
On May 19, 2020, CytoDyn announced it will be coordinating with the National Institution of Health of Mexico…

On May 18, 2020, the NIH reported that roughly 3 to 5% of children with an aunt or…
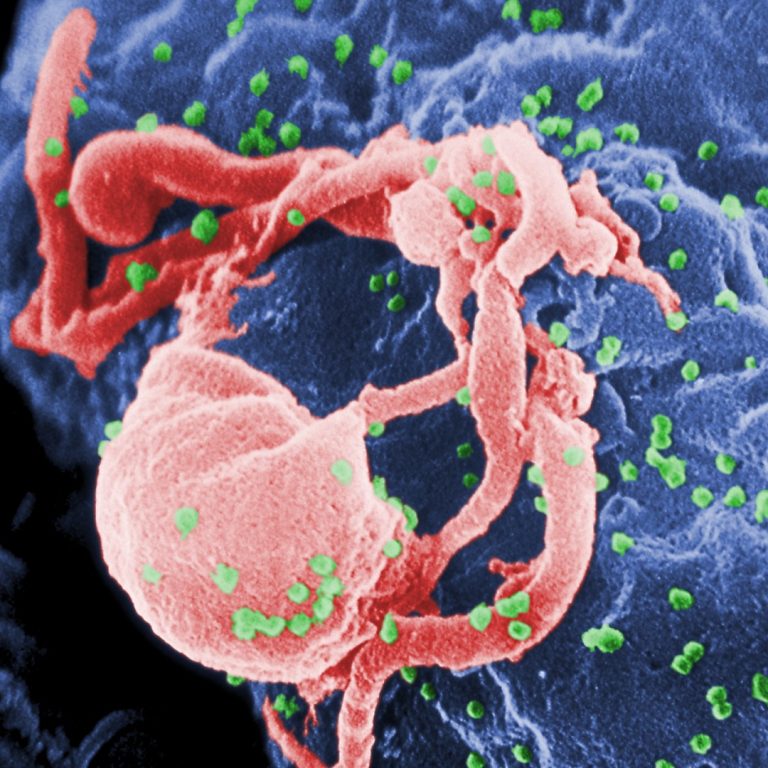
On May 18, 2020, an investigational long-acting form of the HIV drug cabotegravir injected once every 8 weeks…