NIH shifted non-mission-critical laboratory operations to minimal maintenance phase
On Mar. 20, 2020, to further reduce the risk of transmission of COVID-19 and enhance the safety of…

On Mar. 20, 2020, to further reduce the risk of transmission of COVID-19 and enhance the safety of…

On Mar. 20, 2020, BioReference Labs, an OPKO Health company, announced a collaboration with the State of New…
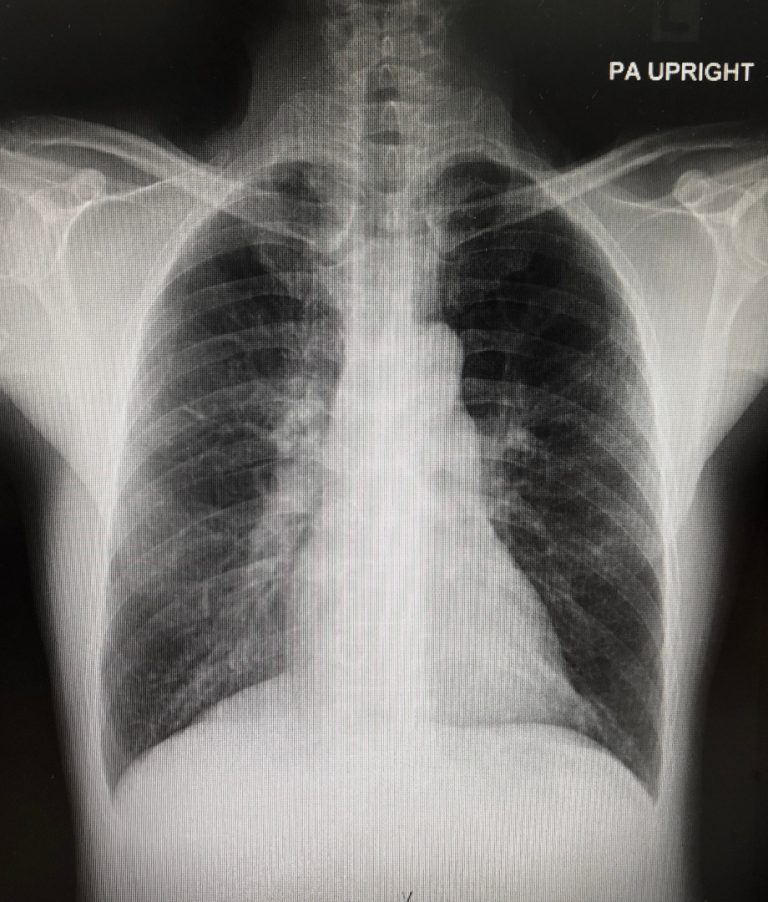
On Mar. 20, 2020, a study was published in JAMA that expands the understanding of influenza-associated complications. Influenza…

On Mar. 19, 2020, Todos Medical announced it had entered into an exclusive distribution agreement for the U.S….

On Mar. 19, 2020, Abbott announced the U.S. Food and Drug Administration (FDA) has issued Emergency Use Authorization (EUA)…
On Mar. 19, 2020, The UArizona Cancer Center announced it was awarded a five-year, $6.9 million prestigious Program…
On Mar. 19, 2020, Gilead Sciences announced the U.S. Food and Drug Administration (FDA) had approved a supplemental…
On Mar. 19, 2020, University of California, Berkeley, investigators have now built such a camera: a microscope that…

On Mar. 19, 2020, Mesa Biotech announced it has significantly expanded its capacity to test clinical samples for…

On Mar. 19, 2020, LabCorp announced it is dedicating the first hour of service each day at its…

On Mar. 18, 2020, StartX, a non-profit startup community of more than 1500 Stanford faculty and alumni founders,…

On Mar. 18, 2020, scientists from the University of Oxford’s Engineering Science Department and the Oxford Suzhou Centre…
On Mar. 18, 2020, Nike CEO John Donahoe and his wife, Eileen, along with Nike Chairman Mark Parker…
On Mar. 17, 2020, scientists reported the novel SARS-CoV-2 coronavirus that emerged in the city of Wuhan, China,…

On Mar. 17, 2020, BioReference Laboratories, an OPKO Health company, announced a collaboration with the New York City…
On Mar. 17, 2020, Todos Medical announced a non-exclusive distribution agreement with 3D Biomedicine Science & Technology, a…

On Mar. 17, 2020, Quidel Corp. announced it had received Emergency Use Authorization (EUA) from the U.S. Food…
On Mar. 17, 2020, MannKind announced it was adjusting research and development resources that were reserved for its…

On Mar. 16, 2020, Hologic announced the U.S. Food and Drug Administration (FDA) had granted Emergency Use Authorization…

On Mar. 16, 2020, BD (Becton, Dickinson and Company) announced the companies had submitted Emergency Use Authorization requests…

On Mar. 15, 2020, Iceland health authorities and deCode Genetics announced they began comprehensive screening for the virus…
On Mar. 15, 2020, the National Institutes of Health (NIH) informed its staff that it had its first…

On Mar. 13, 2020, Roche announced the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization…

On Mar. 13, 2020, Thermo Fisher Scientific announced the U.S. Food and Drug Administration (FDA) has issued an emergency…

On Mar. 13, 2020, declared a COVID-related emergency under Section 501(b) of the Stafford Act [18]. Under the…

On Mar. 13, 2020, IDEXX Laboratories announced the company had seen no positive results in pets to date…

On Mar. 13, 2020, Johnson & Johnson announced that its Janssen Pharmaceutical Companies have entered a collaboration with…

On Mar. 13, 2020, BioReference Laboratories, an OPKO company, announced it had begun accepting specimens for testing of…

On Mar. 13, 2020, Luminex announced that four independent clinical laboratories had validated laboratory developed tests (LDTs) for…

On Mar. 13, 2020, PATH and the Centers for Disease Control and Prevention (CDC) announced a collaboration with…