Bacteria that causes rare disease Melioidosis discovered in U.S. environmental samples
On Jul. 27, 2022, the Centers for Disease Control and Prevention (CDC) announced that it had identified for…

On Jul. 27, 2022, the Centers for Disease Control and Prevention (CDC) announced that it had identified for…
On Jul. 27, 2022, BD (Becton, Dickinson) announced their newly developed molecular polymerase chain reaction (PCR) test for…

On Jul. 27, 2022, Pfizer and BioNTech announced that the companies had initiated a randomized, active-controlled, observer-blind, Phase…
On Jul. 27, 2022, City of Hope announced that a 66-year-old man who was diagnosed with HIV in…

On Jul. 26, 2022, Novavax announced that Nuvaxovid (NVX-CoV2373) COVID-19 vaccine had received expanded manufacturing and marketing approval…

On Jul. 26, 2022, Novavax announced that the Australian Therapeutic Goods Agency had granted expanded approval for provisional…
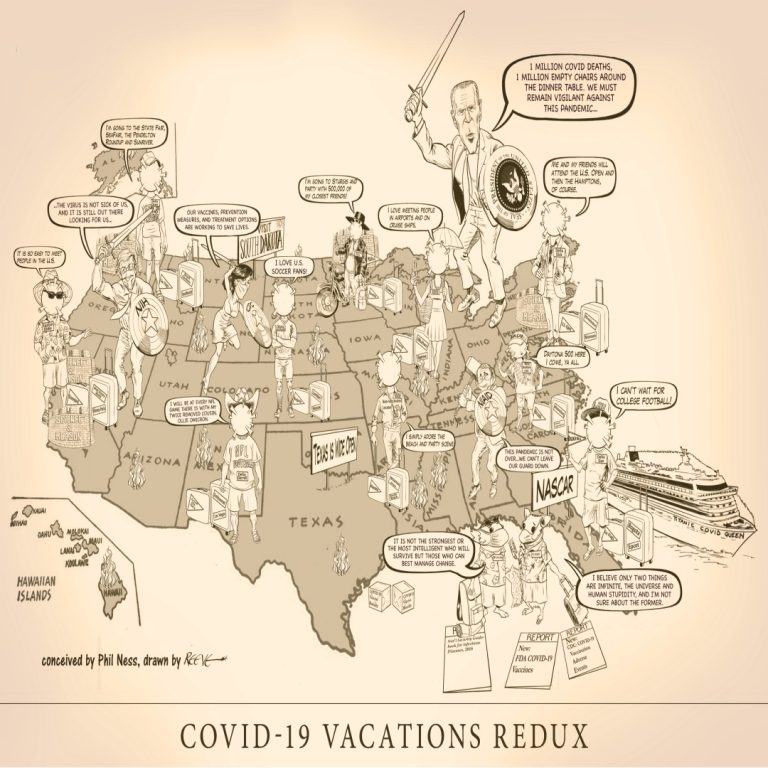
COVID-19 Vacations Redux iIlustrates once again the pits and perils vacation travelers face as they begin their Summer…
On Jul. 22, 2022, Gilead Sciences announced that the Committee for Medicinal Products for Human Use (CHMP) of…
On Jul. 22, 2022, the Centers for Disease Control and Prevention (CDC) released a MMWR report that summarized…

On Jul. 22, 2022, Moderna announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human…

On Jul. 22, 2022, a team of scientists announced they had sequenced the genome of a 6,000-year-old citrullus…

On Jul. 22, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…
On Jul. 21, 2022, The New York State Department of Health confirmed that a case of paralytic poliomyelitis…
On Jul. 19, 2022, Yale scientists announced they had developed a novel Omicron-specific mRNA vaccine that offers superior…

On Jul. 19, 2022, the NIH announced that although COVID-19 booster vaccinations in adults elicit high levels of…

On Jul. 19, 2022, Novavax announced that the U.S. Centers for Disease Control and Prevention’s (CDC) Advisory Committee…
On Jul. 19, 2022, Gilead Sciences and the European Commission announced a new joint procurement agreement (JPA) that…
On Jul. 19, 2022, Novavax announced that it had signed agreements with its partner, SK bioscience, for the…
On Jul. 18, 2022, Moderna announced that the Therapeutic Goods Administration (TGA) in Australia had granted provisional registration…

On Jul. 17, 2022, Ghana announced the preliminary finding of two cases of Marburg virus disease and if…

On Jul. 15, 2022, The largest sustained decline in childhood vaccinations in approximately 30 years has been recorded…

On Jul. 14, 2022, Moderna announced that Health Canada had approved the use of Moderna’s mRNA COVID-19 vaccine,…

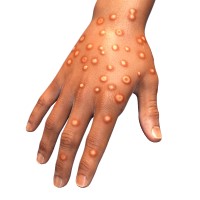
On Jul 14. 2022, Aegis Sciences announced it is was authorized to begin testing for monkeypox using the…

On Jul 13. 2022, Quest Diagnostics announced the nationwide availability of the company’s lab-developed molecular diagnostic test to…
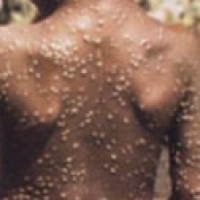
On Jul. 13, 2022, Quest Diagnostics announced that it had announced the nationwide availability* of the company’s lab-developed…

On Jul. 13, 2022, Novavax announced that its COVID-19 Vaccine, Adjuvanted (NVX-CoV2373) had received emergency use authorization from…

On Jul. 11, 2022, Novavax announced an agreement with the U.S. Department of Health and Human Services (HHS),…
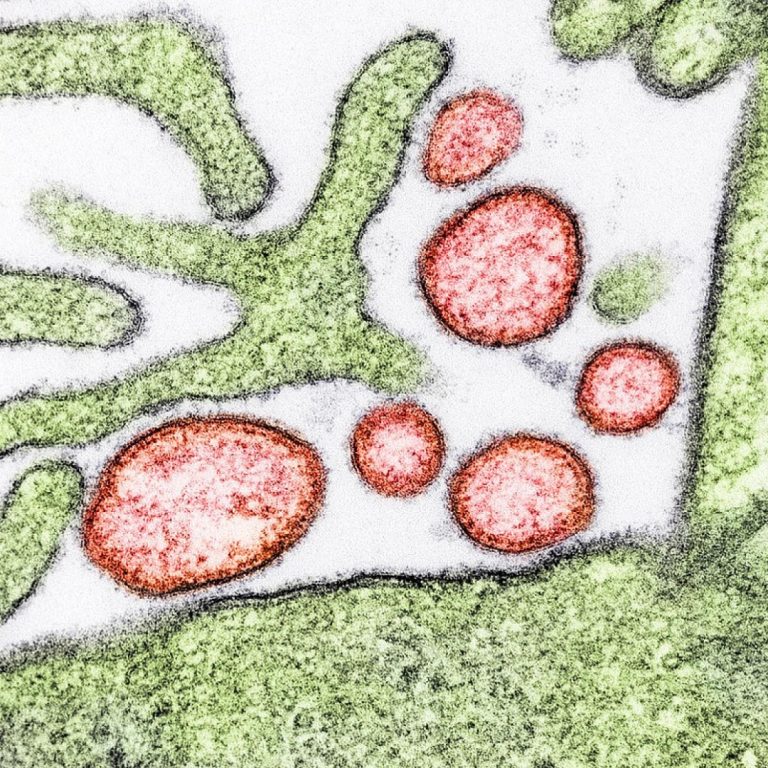
On Jul. 11, 2022, the National Institute of Allergy and Infectious Diseases (NIAID) launched an early-stage clinical trial…

On Jul. 11, 2022, Moderna announced new clinical data on its bivalent Omicron (BA.1) booster candidate, mRNA-1273.214. One…

On Jul. 8, 2022, Pfizer and BioNTech announced that the companies had submitted a variation to the European…